
(a)
Interpretation:
The sign and magnitude of
Concept Introduction :
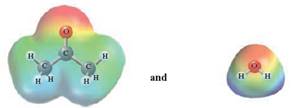
The intermolecular forces in a solute can be broken down by new interactions from the solution as each solute particle will be surrounded by solvent particles in a solution. This is possible when there is disruption between the solute -solute and solvent-solvent interaction.
(a)
Answer to Problem 26E
The values for
Therefore
Explanation of Solution
The process of formation of solution takes place in 3 main steps
- The solutes separating into individual components require energy making it an endothermic reaction.
- The intermolecular forces in solvent must be such that it can make space for solute which requires energy making it an endothermic reaction.
- To allow the solvent and solute molecules to interact which absorbs energy making it an exothermic reaction.
The formation of solution involves enthalpy changes which is depicted as
| Result | |||||
| Non polar solute, Non polar solvent | Small | Small | Small | Small | Solution will form |
| Non-polar solute, polar solvent | Small | Large | Small | Large, positive | No-solution will form |
| Polar solute, Non polar solvent | Large | Small | Small | Large negative | No solution will form |
| Polar solute-polar solvent | Large | Large | Large, negative | small | Solution will form |
In this reaction, acetone reacts with water and as water is polar in nature, the values of each enthalpy changes are depicted as
| Outcome | |||||
| Polar solute, Non polar solvent | Large | Small | Small | Large negative | No solution will form |
(b)
Interpretation:
The sign and magnitude of
Concept Introduction :
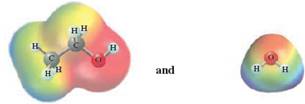
The intermolecular forces in a solute can be broken down by new interactions from the solution as each solute particle will be surrounded by solvent particles in a solution. This is possible when there is disruption between the solute -solute and solvent-solvent interaction.
(b)
Answer to Problem 26E
The values for
Therefore
Explanation of Solution
The process of formation of solution takes place in 3 main steps
- The solutes separating into individual components require energy making it an endothermic reaction.
- The intermolecular forces in solvent must be such that it can make space for solute which requires energy making it an endothermic reaction.
- To allow the solvent and solute molecules to interact which absorbs energy making it an exothermic reaction.
The formation of solution involves enthalpy changes which is depicted as
| Result | |||||
| Non polar solute, Non polar solvent | Small | Small | Small | Small | Solution will form |
| Non-polar solute, polar solvent | Small | Large | Small | Large, positive | No-solution will form |
| Polar solute, Non polar solvent | Large | Small | Small | Large negative | No solution will form |
| Polar solute-polar solvent | Large | Large | Large, negative | small | Solution will form |
In this reaction, ethanol reacts with water and as water is polar in nature, the values of each enthalpy changes are depicted as
| Outcome | |||||
| Polar solute-polar solvent | Large | Large | Large, negative | small | Solution will form |
(c)
Interpretation:
The sign and magnitude of
Concept Introduction :
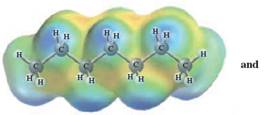
The intermolecular forces in a solute can be broken down by new interactions from the solution as each solute particle will be surrounded by solvent particles in a solution. This is possible when there is disruption between the solute -solute and solvent-solvent interaction.
(c)
Answer to Problem 26E
The values for
Therefore
Explanation of Solution
The process of formation of solution takes place in 3 main steps
- The solutes separating into individual components require energy making it an endothermic reaction.
- The intermolecular forces in solvent must be such that it can make space for solute which requires energy making it an endothermic reaction.
- To allow the solvent and solute molecules to interact which absorbs energy making it an exothermic reaction.
The formation of solution involves enthalpy changes which is depicted as:
| Result | |||||
| Non polar solute, Non polar solvent | Small | Small | Small | Small | Solution will form |
| Non-polar solute, polar solvent | Small | Large | Small | Large, positive | No-solution will form |
| Polar solute, Non polar solvent | Large | Small | Small | Large negative | No solution will form |
| Polar solute-polar solvent | Large | Large | Large, negative | small | Solution will form |
In this reaction, heptane reacts with hexane and as both is non-polar in nature; the values of each enthalpy changes are depicted as:
| Outcome | |||||
| Non polar solute, Non polar solvent | Small | Small | Small | Small | Solution will form |
(d)
Interpretation:
The sign and magnitude of
Concept Introduction :
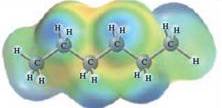
The intermolecular forces in a solute can be broken down by new interactions from the solution as each solute particle will be surrounded by solvent particles in a solution. This can happen when there is disruption between the solute-solute and solvent-solvent interaction.
(d)
Answer to Problem 26E
The values for
Therefore
Explanation of Solution
The process of formation of solution takes place in 3 main steps
- The solutes separating into individual components require energy making it an endothermic reaction.
- The intermolecular forces in solvent must be such that it can make space for solute which requires energy making it an endothermic reaction.
- To allow the solvent and solute molecules to interact which absorbs energy making it an exothermic reaction.
The formation of solution involves enthalpy changes which are depicted as:
| Result | |||||
| Non polar solute, Non polar solvent | Small | Small | Small | Small | Solution will form |
| Non-polar solute, polar solvent | Small | Large | Small | Large, positive | No-solution will form |
| Polar solute, Non polar solvent | Large | Small | Small | Large negative | No solution will form |
| Polar solute-polar solvent | Large | Large | Large, negative | small | Solution will form |
In this reaction, heptane reacts with water wherein heptane is non-polar and water is polar in nature. The values of each enthalpy changes are depicted as:
| Result | |||||
| Non-polar solute, polar solvent | Small | Large | Small | Large, positive | Solution will not form. |
Want to see more full solutions like this?
Chapter 17 Solutions
Chemical Principles
- Which solute has the greatest effect on the boiling pointof 1.00 kg of water: 50.0 g of strontium chloride (SrCl2) or 150.0 g of carbon tetrachloride (CCl4) ? Justify youranswer.arrow_forwardThe dispersed phase of a certain colloidal dispersion consists of spheres of diameter 1.0 102 nm. (a) What are the volume (V=43r2) and surface area (A = r2) of each sphere? (b) How many spheres are required to give a total volume of 1.0 cm3? What is the total surface area of these spheres in square meters?arrow_forwardWhat is the freezing point and normal boiling point of a solution made by adding 39 mL of acetone, C3H6O, to 225 mL of water? The densities of acetone and water are 0.790 g/cm3 and 1.00 g/cm3, respectively.arrow_forward
- 6-111 As noted in Section 6-8C, the amount of external pressure that must be applied to a more concentrated solution to stop the passage of solvent molecules across a semipermeable membrane is known as the osmotic pressure The osmotic pressure obeys a law similar in form to the ideal gas law (discussed in Section 5-4), where Substituting for pressure and solving for osmotic pressures gives the following equation: RT MRT, where M is the concentration or molarity of the solution. (a) Determine the osmotic pressure at 25°C of a 0.0020 M sucrose (C12H22O11) solution. (b) Seawater contains 3.4 g of salts for every liter of solution. Assuming the solute consists entirely of NaCl (and complete dissociation of the NaCI salt), calculate the osmotic pressure of seawater at 25°C. (c) The average osmotic pressure of blood is 7.7 atm at 25°C. What concentration of glucose (C6H12O6) will be isotonic with blood? (d) Lysozyme is an enzyme that breaks bacterial cell walls. A solution containing 0.150 g of this enzyme in 210. mL of solution has an osmotic pressure of 0.953 torr at 25°C. What is the molar mass of lysozyme? (e) The osmotic pressure of an aqueous solution of a certain protein was measured in order to determine the protein's molar mass. The solution contained 3.50 mg of protein dissolved in sufficient water to form 5.00 mL of solution. The osmotic pressure of the solution at 25°C was found to be 1.54 torr. Calculate the molar mass of the protein.arrow_forwardCarbon tetrachloride (CCl4) and benzene (C6H6) form ideal solutions. Consider an equimolar solution of CCl4 and C6H6 at 25C. The vapor above the solution is collected and condensed. Using the following data, determine the composition in mole fraction of the condensed vapor. Substance Gfo C6H6(l) 124.50 kJ/mol C6H6(g) 129.66 kJ/mol CCI4(l) 65.21 kJ/mol CCI4,(g) 60.59 kJ/molarrow_forwardThe Rast method uses camphor (C10H16O) as a solvent for determining the molar mass of a compound. When 2.50 g of cortisone acetate are dissolved in 50.00 g of camphor (k f =40.0C/m), the freezing point of the mixture is 173.44C; that of pure camphor is 178.40C. What is the molar mass of cortisone acetate?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





