
Physics (5th Edition)
5th Edition
ISBN: 9780321976444
Author: James S. Walker
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 41PCE
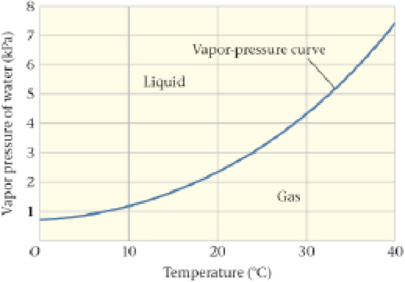
Vapor Pressure for Water Figure 17-35 shows a portion of the vapor-pressure curve for water. Referring to the figure, estimate the pressure that would be required for water to boil at 30 °C.
Figure 17-35 Problems 41, 42, 43, and 44
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An aluminum cup of 100 cm^3 capacity is filled with glycerin at 23 degrees Celsius. How much glycerin, if any, will spill out of the cup if the temperature of the cup and glycerin is raised to 28 degrees Celsius? (The coefficient of volume expansion of glycerin is 5.1×10^(-4)/degree Celsius.)
A student walking to class on a cold day (To = 4 °C) find a silver ring with an inner diameter of D = 1.85 cm. The silver has a coefficient of linear expansion of α = 18.6 × 10-6 1/C.
a. Input an expression for the rings inner diameter Dh when the student warms it to their body temperature, Tb.
b. What is the change in diameter, in units of millimeters, if Tb = 37 °C?
A spherical steel ball bearing has a diameter of 2.540 cm at 26.00°C. (Assume the coefficient of linear expansion of the steel is 1.10 10-5(°C-1), and the average volume expansion coefficient is 3.30 10-5(°C-1).) A) What is its diameter when its temperature is raised to 97.0°C? (Give your answer to four significant figures in cm.) B) What temperature change is required to increase its volume by 1.100%?
Chapter 17 Solutions
Physics (5th Edition)
Ch. 17.1 - Rank the following ideal-gas systems in order of...Ch. 17.2 - If the Kelvin temperature of a gas is doubled, by...Ch. 17.3 - A metal rod of a given initial length and...Ch. 17.4 - A portion of a substances phase diagram is shown...Ch. 17.5 - Which requires more heat: melting 100 kg of copper...Ch. 17.6 - An ice cube is placed in a cup of water. A few...Ch. 17 - How is the air pressure in a tightly sealed house...Ch. 17 - The average speed of air molecules in your room is...Ch. 17 - Is it possible to change both the pressure and the...Ch. 17 - Prob. 4CQ
Ch. 17 - A camping stove just barely boils water on a...Ch. 17 - An autoclave is a device used to sterilize medical...Ch. 17 - As the temperature of ice is increased, it changes...Ch. 17 - BIO Isopropyl alcohol is sometimes rubbed onto a...Ch. 17 - A drop of water on a kitchen counter evaporates in...Ch. 17 - (a) Is the number of molecules in one mole of N2...Ch. 17 - Predict/Explain If you put a helium-filled balloon...Ch. 17 - Two containers hold ideal gases at the same...Ch. 17 - Prob. 4PCECh. 17 - BIO After emptying her lungs, a person inhales 4.3...Ch. 17 - An automobile tire has a volume of 0.0185 m3. At a...Ch. 17 - Prob. 7PCECh. 17 - A compressed-air tank holds 0.500 m3 of air at a...Ch. 17 - Four ideal gases have the following pressures, P,...Ch. 17 - A balloon contains 3.9 liters of nitrogen gas at a...Ch. 17 - Prob. 11PCECh. 17 - Predict/Calculate A bicycle tire with a volume of...Ch. 17 - A 515-cm3 flask contains 0.460 g of a gas at a...Ch. 17 - Prob. 14PCECh. 17 - The air inside a hot-air balloon has an average...Ch. 17 - Prob. 16PCECh. 17 - Consider the system described in the previous...Ch. 17 - Prob. 18PCECh. 17 - Prob. 19PCECh. 17 - If the translational speed of molecules in an...Ch. 17 - At what temperature is the rms speed of H2 equal...Ch. 17 - Suppose a planet has an atmosphere of pure ammonia...Ch. 17 - Prob. 23PCECh. 17 - Prob. 24PCECh. 17 - Prob. 25PCECh. 17 - What is the temperature of a gas of CO2 molecules...Ch. 17 - The rms speed of a sample of gas is increased by...Ch. 17 - Prob. 28PCECh. 17 - A 380-mL spherical flask contains 0.065 mol of an...Ch. 17 - Prob. 30PCECh. 17 - A rock climber hangs freely from a nylon rope that...Ch. 17 - BIO To stretch a relaxed biceps muscle 2.5 cm...Ch. 17 - A 22-kg chimpanzee hangs from the end of a...Ch. 17 - The Marianas Trench The deepest place in all the...Ch. 17 - Four cylindrical rods with various cross-sectional...Ch. 17 - Predict/Calculate A steel wire 4.1 m long...Ch. 17 - BIO Spiderweb An orb weaver spider with a mass of...Ch. 17 - Predict/Calculate Two rods of equal length (0.55...Ch. 17 - A piano wire 0.82 m long and 0.93 mm in diameter...Ch. 17 - The formation of ice from water is accompanied by...Ch. 17 - Vapor Pressure for Water Figure 17-35 shows a...Ch. 17 - Using the vapor-pressure curve given in Figure...Ch. 17 - Prob. 43PCECh. 17 - Prob. 44PCECh. 17 - Predict/Calculate The Vapor Pressure of CO2 A...Ch. 17 - Phase Diagram for Water The phase diagram for...Ch. 17 - Phase Diagram for CO2 The phase diagram for CO2 is...Ch. 17 - Prob. 48PCECh. 17 - How much heat must be removed from 1.96 kg of...Ch. 17 - A heat transfer of 9.5 105 J is required to...Ch. 17 - How much heat must be added to 2.55 kg of copper...Ch. 17 - An ammonia refrigeration cycle involves the...Ch. 17 - Prob. 53PCECh. 17 - Prob. 54PCECh. 17 - Prob. 55PCECh. 17 - Figure 17-30 shows a temperature-versus-heat plot...Ch. 17 - Predict/Calculate Suppose the 1.000 kg of water in...Ch. 17 - Prob. 58PCECh. 17 - When you go out to your car one cold winter...Ch. 17 - A large punch bowl holds 3.99 kg of lemonade...Ch. 17 - A 155-g aluminum cylinder is removed from a liquid...Ch. 17 - An 825-g iron block is heated to 352 C and placed...Ch. 17 - Party Planning You are expecting to serve 32 cups...Ch. 17 - Predict/Calculate A 35-g ice cube at 0.0 C is...Ch. 17 - A 48-g block of copper at 12 C is added to 110 g...Ch. 17 - A 0 075-kg ice cube at 0.0 C is dropped into a...Ch. 17 - To help keep her barn warm on cold days, a farmer...Ch. 17 - CE As you go up in attitude, do you expect the...Ch. 17 - Prob. 69GPCh. 17 - Prob. 70GPCh. 17 - Prob. 71GPCh. 17 - Cooling Computers Researchers are developing heat...Ch. 17 - Prob. 73GPCh. 17 - Prob. 74GPCh. 17 - Evaporating Atmosphere Hydrogen gas evaporates...Ch. 17 - Prob. 76GPCh. 17 - A Boiling Geyser (a) The column of water that...Ch. 17 - A Melting Glacier (a) A glacier is made of ice of...Ch. 17 - Peter catches a 4 2-kg striped bass on a fishing...Ch. 17 - A steel ball (density=7860kg/m3) with a diameter...Ch. 17 - A lead brick with the dimensions shown in Figure...Ch. 17 - (a) Find the amount of heat that must be extracted...Ch. 17 - Mighty Ice Lift A tremendous force is generated...Ch. 17 - Orthopedic Implants Metals such as titanium and...Ch. 17 - Students on a spring break picnic bring a cooler...Ch. 17 - A 5.9-kg block of ice at 1.5 C slides on a...Ch. 17 - A cylindrical copper rod 37 cm long and 7.5 cm in...Ch. 17 - Prob. 88PPCh. 17 - Prob. 89PPCh. 17 - Prob. 90PPCh. 17 - Prob. 91PPCh. 17 - Referring to Example 17-17 (a) Find the final...Ch. 17 - Referring to Example 17-17 (a) Find the final...
Additional Science Textbook Solutions
Find more solutions based on key concepts
1. How many significant figures does each of the following numbers have?
a. 0.73 b. 7.30 c. 73 d. 0.073
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Explain all answers clearly, with complete sentences and proper essay structure if needed. An asterisk (*) desi...
The Cosmic Perspective Fundamentals (2nd Edition)
A thick ring of mass M has inner radius R1 and outer radius R2. Show that its rotational inertia is given by 12...
Essential University Physics (3rd Edition)
48. To make your workouts more productive, you can get an electrical generator that you drive with the rear whe...
College Physics: A Strategic Approach (4th Edition)
A mole is approximately the number of protons in a gram of protons. The mass of a neutron is about the same as ...
An Introduction to Thermal Physics
24. (Il) You buy a plastic dart gun, and being a clever physics student you decide to do a quick calculation to...
Physics: Principles with Applications
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- If the average kinetic energy of the molecules in an ideal gas initially at 20C doubles, what is the final temperature of the gas? (5.6) (a) 10C (b) 40C (c) 313C (d) 586Carrow_forwardThe average coefficient of linear expansion of copper is 17 106 (C)1. The Statue of Liberty is 93 in tall on a summer morning when the temperature is 25C. Assume the copper plates covering the statue are mounted edge to edge without expansion joints and do not buckle or bind on the framework supporting them as the day grows hot. What is the order of magnitude of the statues increase in height? (a) 0.1 mm (b) 1 mm (c) 1 cm (d) 10 cm (e) 1 marrow_forwardThe pressure gauge on a cylinder of gas registers the gauge pressure, which is the difference between the interior pressure and the exterior pressure P0. Lets call the gauge pressure Pg. When the cylinder is full, the mass of the gas in it is mi at a gauge pressure of Pgi. Assuming the temperature of the cylinder remains constant, show that the mass of the gas remaining in the cylinder when the pressure reading is Pgf is given by mf=mi(Pgf+P0Pgi+P0)arrow_forward
- A 5.00-L vessel contains nitrogen gas at 27.0C and 3.00 atm. Find (a) the total translational kinetic energy of the gas molecules and (b) the average kinetic energy per molecule.arrow_forwardThe vapor pressure of water at 40.0ºC is 7.34×103 N/m2. Using the ideal gas law, calculate the density of water vapor in g/m3 that creates a partial pressure equal to this vapor pressure. The result should be the same as the saturation vapor density at that temperature (51.1 g/m3 ).arrow_forwardBefore leaving on a trip to Melbourne Beach, Florida, you measure the pressure inside the tires of your car at a gas station. At – 7.5 C (chilly!), the tire pressure is 206.5 kPa. When you arrive in Florida, you check the tire pressure and find it is now 236.5 kPa. What is the temperature in Melbourne Beach?arrow_forward
- Liquid water turns out to have a higher density at4 Degrees Celcius than at any other temperature.What does this tell us about the coefficient of thermal expansion of water between 0 and4 degrees?(A) It is negative.(B) It is zero.(C) It is positive but somewhat small.(D) It is positive and very large.arrow_forwardSuppose that air in the human lungs has a temperature of 37°C, and the partial pressure of water vapor has a value of 5.5 x 103 Pa. What is the relative humidity in the lungs? Consult the vapor pressure curve for water that accompanies this problem. Express your answer as a percentage with no units.arrow_forwardThe George Washington Bridge crosses the Hudson River between New York and New Jersey. The span of the steel bridge is about 1.90 km. What might be the difference in meters between the maximum span and minimum span over the course of a year, if the temperature can vary from a low of −(5.4x10^0)°C in winter to a high of 42°C in summer? Expansion coefficient of steel is α = 12.0 × 10−6 K−1. Use two significant figures in your answer.arrow_forward
- Problem 13: A bicycle tire has a pressure of 6.8 × 105 Pa at a temperature of 15°C and contains 2.00 L of gas. What will its pressure be, in pascals, if you let out an amount of air that has a volume of 115 cm3 at atmospheric pressure and at the temperature of the tire? Assume tire temperature and volume remain constant.arrow_forwardLiquid water turns out to have a higher density at4C degrees than at any other temperature. What does this tell us about the coefficient of thermal expansion of water between 0 and 4 degrees?(A) It is negative.(B) It is zero.(C) It is positive but somewhat small.(D) It is positive and very large.arrow_forwardA student walking to class on a cold day (To = 1 °C) find a silver ring with an inner diameter of D = 1.75 cm. The silver has a coefficient of linear expansion of α = 18.3 × 10-6 1/C. Input an expression for the rings inner diameter Dh when the student warms it to their body temperature, Tb and calculate the change in diameter in mm if Tb = 37 °C? ΔD = 0.635 is incorrectarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY