
Concept explainers
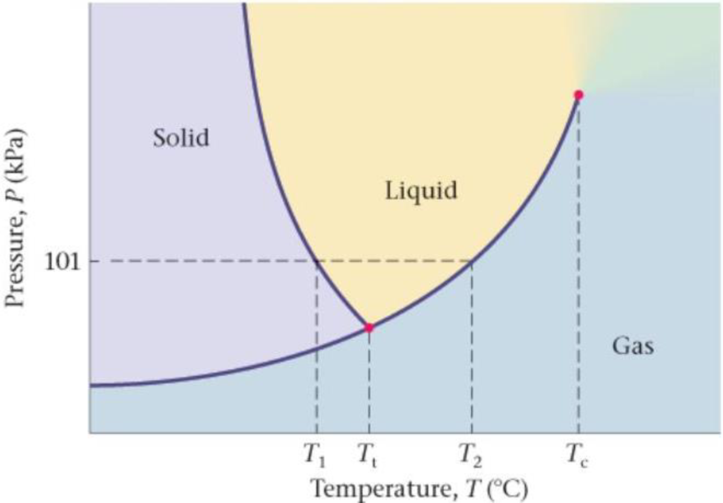
Phase Diagram for Water The phase diagram for water is shown in Figure 17-37. (a) What is the temperature T1 on the phase diagram? (b) What is the temperature T2 on the phase diagram? (c) What happens to the melting/freezing temperature of water if atmospheric pressure is decreased? Justify your answer by referring to the phase diagram (d) What happens to the boiling/condensation temperature of water if atmospheric pressure is increased? Justify your answer by referring to the phase diagram.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Physics (5th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Essential University Physics (3rd Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Tutorials in Introductory Physics
University Physics (14th Edition)
Introduction to Electrodynamics
- What is the vapor pressure of solid carbon dioxide (dry ice) at _785 Figure 13.36 The phase diagram for carbon dioxide. The axes aha nonlinear, and the graph is not to scale. Dry ice is solid carbon dioxide and has a sublimation temperature ofarrow_forwardIf you add boiling water to a cup at room temperature, what would you expect the final equilibrium temperature of the unit to be? You will need to include 1he surroundings as pan of the system. Consider the zeroth law of thermodynamics.arrow_forward(a) If tossing 100 coins, how many ways (microstates) are there to get me three most likely macro states of 49 heads and 51 tails, 50 heads and 50 tails, and 51 heads and 49 tails? (b) What percent of the total possibilities is this? (Consult Table 15.4.)arrow_forward
- Suppose a gasfilled incandescent light bulb is manufactured so that the gas inside the bulb is at atmospheric pressure when the bulb has a temperature of 20.0C. (a) Find the gauge pressure inside such a bulb when it is hot, assuming its average temperature is 60.0C (an approximation) and neglecting any change in volume due to thermal expansion or gas leaks. (b) The actual final pressure for the light bulb will be less than calculated in part (a) because the glass bulb will expand. What will the actual final pressure be, taking this into account? Is this a negligible difference?arrow_forward(II) You have a sample of water and are able to control temperature and pressure arbitrarily. (a) Using Fig. 13–22, describe the phase changes you would see if you started at a temperature of 85°C, a pressure of 180 atm, and decreased the pressure down to 0.004 atm while keeping the temperature fixed. (b) Repeat part (a) with the temperature at 0.0°C. Assume that you held the system at the starting conditions long enough for the system to stabilize before making further changes.arrow_forwardThe steak span of the George Washington Bridge is about 1.6 km at -26.0 C. How long is the bridge on a hot summer day when the temperature is 40.6 C? I added a section of the question in the picture Thank you!arrow_forward
- Given the following measurements of mean winds and temperature from a 200-m mast in Netherlands on a September night: Table 1. Measurements of mean winds and temperature from a 200-m mast in Netherlands on a September night. Question: Determine the potential temperature gradient (∂Θ/∂z) at heights of 7.5, 15, 30, 60, 100, 140, and 180 m (note: ∂Θ/∂z at height 7.5 m is the potential temperature gradient between 5 m and 10 m).arrow_forward10. If it takes 554 calories to increase the temperature of 100 grams of a substance by 60.0 degrees Celsius then how many calories does it take to raise the temperature of 600 grams of the same substance by 60.0 degrees Celsius? No phase changes occur. a. 13300 b. 554 c. 3324arrow_forwardTwo metal bars are made of invar and a third bar is made of aluminum. At 0°C, each of the three bars is drilled with two holes 40.0 cm apart. Pins are put through the holes to assemble the bars into an equilateral triangle as shown. (a) First ignore the expansion of the invar. Find the angle between the invar bars as a function of Celsius temperature. (b) Is your answer accurate for negative as well as positive temperatures? (c) Is it accurate for 0°C? (d) Solve the problem again, including the expansion of the invar. Aluminum melts at 660°C and invar at 1 427°C. Assume the tabulated expansion coefficients are constant. What are (e) the greatest and (f) the smallest attainable angles between the invar bars?arrow_forward
- Draw a diagram (T-V) in the same molar composition as well as a P-xy diagram at 25 ° C and a T-xy diagram at atmospheric pressure for the toluene-cyclohexane system. Hello, solve this question for me in Type ( not handwriting)arrow_forwardUsing Table 16-2, how many Joules of heat does it take to increase the temperature of one kilogram of water by TWO degrees Kelvin (or two degrees Celsius for that matter). Remember a delta T in Celsius is the same delta T in Kelvin because both temperature scales experience the same magnitude changes in temperature.arrow_forwardTwo objects (A and B) are placed in thermal contact and exchange heat. Object A is initiallyhotter than object B. Some additional information about them is given below. Which best describesthe final equilibrium temperature of the objects? a) between TA and TB but closer to TAb) between TA and TB but closer to TBc) exactly halfway between TA and TBd) need to know the temperatures to answerarrow_forward
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





