
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 17, Problem 48P
Interpretation Introduction
Interpretation:
The
Concept introduction:
The value of
The compound attached by electro withdrawing will have high tendency of protonation, so the compound have high acidic strength.
If the acidity or easiness of the protonation is high for a compound, then the value of
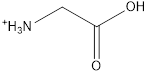
The structure of glycine is,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Glycine has pKa values of 2.3 and 9.6. Would you expect the pKa values of glycylglycine to be higher or lower than these values?
glycine has pka value of 2.3 and 9.6 would you except the pka value of glycine to be higher or lower than these value?
Glycine has pKa values of 2.34 and 9.60. At what pH does glycine exist in the indicated form?
Chapter 17 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 17.1 - a. Explain why, when the imidazole ring of...Ch. 17.2 - Prob. 2PCh. 17.3 - Prob. 3PCh. 17.3 - Prob. 4PCh. 17.3 - Prob. 6PCh. 17.4 - Calculate the pI of each of the following amino...Ch. 17.4 - a. Which amino acid has the lowest pI value? b....Ch. 17.5 - What aldehyde is formed when valine is treated...Ch. 17.5 - Prob. 10PCh. 17.5 - Prob. 11P
Ch. 17.5 - Prob. 12PCh. 17.6 - Prob. 13PCh. 17.6 - What amino acid would be formed using the...Ch. 17.6 - What amino acid would be formed when the aldehyde...Ch. 17.7 - Pig liver esterase is an enzyme that catalyzes the...Ch. 17.8 - Prob. 17PCh. 17.8 - Prob. 18PCh. 17.8 - Prob. 19PCh. 17.8 - Prob. 20PCh. 17.10 - Prob. 21PCh. 17.10 - Prob. 22PCh. 17.10 - Why does cyanogen bromide not cleave on the C-side...Ch. 17.10 - Prob. 24PCh. 17.10 - Prob. 26PCh. 17.12 - Prob. 27PCh. 17.13 - a. Which would have the greatest percentage of...Ch. 17 - Draw the predominant form of the following amino...Ch. 17 - What is the pI of serine?Ch. 17 - Prob. 31PCh. 17 - Prob. 32PCh. 17 - Which would have a higher percentage of negative...Ch. 17 - Draw the form of aspartate that predominates at...Ch. 17 - Prob. 35PCh. 17 - A professor was preparing a manuscript for...Ch. 17 - a. Why is the pKa of the glutamate side chain...Ch. 17 - Prob. 38PCh. 17 - Determine the amino acid sequence of a polypeptide...Ch. 17 - Prob. 40PCh. 17 - Prob. 41PCh. 17 - Three peptides were obtained from a trypsin...Ch. 17 - Prob. 43PCh. 17 - After the polypeptide shown here was treated with...Ch. 17 - The disulfide bridges of a polypeptide were...Ch. 17 - -Amino acids can be prepared by treating an...Ch. 17 - Reaction of a polypeptide with carboxypeptidase A...Ch. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Show how valine can be prepared by a. a Strecker...Ch. 17 - Prob. 51PCh. 17 - Why is proline never found in an -helix?Ch. 17 - Determine the amino acid sequence of a polypeptide...Ch. 17 - Prob. 55PCh. 17 - A chemist wanted to test his hypothesis that the...Ch. 17 - A normal polypeptide and a mutant of the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- glycine has pKa value of 2.3 and 9.6 would you expect pKa value of glyclglycine to be higher to lower than these values?arrow_forwardWhat is the isoelectric point for the peptide MNPQR at pH 7? Draw the structure, including the stereochemistry of each C-alpha chiral center.arrow_forwardThe configuration of the chiral center in a-amino acids is most commonly specified using the d,l convention. It can also be identified using the R,S convention . Does the chiral center in l-serine have the R or S configuration?arrow_forward
- how to determine net charge of amino acid, given pH and pKa values?arrow_forwardPka for terazosin ? With illustrationarrow_forwardCalculate the isoelectric point for each amino acid.a. cysteine: pKa (COOH) = 2.05; pKa (a-NH3+) = 10.25b. methionine: pKa (COOH) = 2.28; pKa (a-NH3+) = 9.21arrow_forward
- The pKa values of each group are shown above. In the structures below, fill in the missing atoms/charges for the amino acid at each indicated pH.arrow_forward1. A given glycine solution was titrated against NaOH and pH of the final solution was 4.8. Given that pKa1 and pKa2 of glycine are 2.34 and 9.60, respectively. Calculate the concentration of the dissociated amino acid in terms of the undissociated amino acid, if [x] and [y] are the concentrations of dissociated and undissociated amino acid, respectively.arrow_forwardAssign an R or S configuration to the chiral center in each amino acid. (a) l-Phenylalanine (b) l-Glutamic acid (c) l-Methioninearrow_forward
- Alanine has pKa values of 2.34 and 9.69. Therefore, alanine exists predominately as a zwitterion in anaqueous solution with pH 7 ____ and pH 6 ____.arrow_forwardChemistry 1a. The lack of an asymmetric carbons and ease of rotation make glycine rich protein sequences: a. Fibrous b. Helical c. Cyclic d. Flexible e. Globular 1b. Introducing D-amino acids into peptides can be useful as a strategy to: a. Change pKa b. Reduce thermal stability c. Alter ionic strength d. Slow degradation e. Increase solubility 1c. Cellulose is not useful dietary carbohydrate because humans: a. Don’t have sufficient non-mitochondrial ATP b. Lack a 1,4-beta glycosidase c. Can’t digest branched chain carbohydrates d. Loose lactase as they mature e. Can’t absorb pentise subunitsarrow_forwardWhy is the side chain Pka of amino acid so much higher ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY