
Basic Chemistry (5th Edition)
5th Edition
ISBN: 9780134138046
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17.1, Problem 17.1QAP
LEARNING GOAL Write the IUPAC names and draw the condensed or line-angle structural formulas for
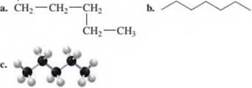
17.1 Write the IUPAC name for each of the following alkanes:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
All part provide compound name plz thank you plz IUPAC and normal
Cyclohexene has a boiling point higher than water.
Group of answer choices
True
False
Only answer B. Illustration.B. Illustration. For each number, draw the structure of the IUPAC named compound to its given space per column. You can use any type of structural formula EXCEPT skeletal structural formula.Kindly make the answers clear and readable.
Chapter 17 Solutions
Basic Chemistry (5th Edition)
Ch. 17.1 - LEARNING GOAL Write the IUPAC names and draw the...Ch. 17.1 - Prob. 17.2QAPCh. 17.1 - Prob. 17.3QAPCh. 17.1 - Prob. 17.4QAPCh. 17.1 - Prob. 17.5QAPCh. 17.1 - Prob. 17.6QAPCh. 17.1 - Prob. 17.7QAPCh. 17.1 - Prob. 17.8QAPCh. 17.1 - Prob. 17.9QAPCh. 17.1 - Prob. 17.10QAP
Ch. 17.2 - Prob. 17.11QAPCh. 17.2 - Prob. 17.12QAPCh. 17.2 - Prob. 17.13QAPCh. 17.2 - Prob. 17.14QAPCh. 17.2 - Prob. 17.15QAPCh. 17.2 - Prob. 17.16QAPCh. 17.2 - Prob. 17.17QAPCh. 17.2 - Prob. 17.18QAPCh. 17.2 - Prob. 17.19QAPCh. 17.2 - Prob. 17.20QAPCh. 17.2 - Prob. 17.21QAPCh. 17.2 - Prob. 17.22QAPCh. 17.2 - Prob. 17.23QAPCh. 17.2 - Prob. 17.24QAPCh. 17.3 - Prob. 17.25QAPCh. 17.3 - Prob. 17.26QAPCh. 17.3 - Prob. 17.27QAPCh. 17.3 - Prob. 17.28QAPCh. 17.4 - Prob. 17.29QAPCh. 17.4 - Prob. 17.30QAPCh. 17.4 - Prob. 17.31QAPCh. 17.4 - Prob. 17.32QAPCh. 17.4 - Prob. 17.33QAPCh. 17.4 - Prob. 17.34QAPCh. 17.5 - Prob. 17.35QAPCh. 17.5 - Prob. 17.36QAPCh. 17.5 - Prob. 17.37QAPCh. 17.5 - Prob. 17.38QAPCh. 17.5 - Prob. 17.39QAPCh. 17.5 - Prob. 17.40QAPCh. 17.6 - Prob. 17.41QAPCh. 17.6 - Prob. 17.42QAPCh. 17.6 - Prob. 17.43QAPCh. 17.6 - Prob. 17.44QAPCh. 17.6 - Prob. 17.45QAPCh. 17.6 - Prob. 17.46QAPCh. 17.6 - Prob. 17.47QAPCh. 17.6 - Prob. 17.48QAPCh. 17.6 - Prob. 17.49QAPCh. 17.6 - Prob. 17.50QAPCh. 17.6 - Prob. 17.51QAPCh. 17.6 - Draw the condensed structural formula for each of...Ch. 17.7 - Write the common name for each of the following:Ch. 17.7 - Prob. 17.54QAPCh. 17.7 - Prob. 17.55QAPCh. 17.7 - Prob. 17.56QAPCh. 17.7 - Prob. 17.57QAPCh. 17.7 - Prob. 17.58QAPCh. 17.7 - Prob. 17.59QAPCh. 17.7 - Prob. 17.60QAPCh. 17 - Prob. 17.61FUCh. 17 - 17.62 New polymers have been synthesized to...Ch. 17 - Prob. 17.63FUCh. 17 - Prob. 17.64FUCh. 17 - Prob. 17.65UTCCh. 17 - Prob. 17.66UTCCh. 17 - Prob. 17.67UTCCh. 17 - Prob. 17.68UTCCh. 17 - Prob. 17.69AQAPCh. 17 - Prob. 17.70AQAPCh. 17 - Prob. 17.71AQAPCh. 17 - Prob. 17.72AQAPCh. 17 - Prob. 17.73AQAPCh. 17 - Prob. 17.74AQAPCh. 17 - Prob. 17.75AQAPCh. 17 - Prob. 17.76AQAPCh. 17 - Prob. 17.77AQAPCh. 17 - Prob. 17.78AQAPCh. 17 - Prob. 17.79AQAPCh. 17 - Prob. 17.80AQAPCh. 17 - Prob. 17.81AQAPCh. 17 - Prob. 17.82AQAPCh. 17 - Prob. 17.83AQAPCh. 17 - Prob. 17.84AQAPCh. 17 - Prob. 17.85AQAPCh. 17 - Prob. 17.86AQAPCh. 17 - Prob. 17.87AQAPCh. 17 - Prob. 17.88AQAPCh. 17 - Prob. 17.89AQAPCh. 17 - Prob. 17.90AQAPCh. 17 - Prob. 17.91AQAPCh. 17 - Prob. 17.92AQAPCh. 17 - Draw the condensed structural formula for each of...Ch. 17 - Prob. 17.94AQAPCh. 17 - Prob. 17.95AQAPCh. 17 - Prob. 17.96AQAPCh. 17 - Prob. 17.97CQCh. 17 - Prob. 17.98CQCh. 17 - Prob. 17.99CQCh. 17 - Prob. 17.100CQCh. 17 - Prob. 17.101CQCh. 17 - Prob. 17.102CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Carboxylic acids are classified according to the substituent that is bonded to the carboxyl carbon into Aliphatic acids and An aromatic acid. Select the correct answer: a. False b. Truearrow_forwardWhat is the preferred IUPAC name of the compound in Figure 12. [Numbers are separated by commas and numbers and letters by hyphens. Use lowercase letters and do not use spaces.] * Your answerarrow_forwardDetermine which compounds show cis-trans isomerism. Draw and label the isomers, using both the cis-trans and E-Znomenclatures where applicable.(a) pent-1-ene (b) pent-2-ene(c) hex-3-enearrow_forward
- Answer choices for question 2: a. rate=k[alkane] b. rate=k[H+] c.rate=k[alkane][H+] d.rate=k[alkane][H2O] Question 3 is asking about the green arrows.arrow_forwardAnswer B. Illustration only.For each number, draw the structure of the IUPAC named compound to its given space per column. You can use any type of structural formula EXCEPT skeletal structural formula.Kindly make the answers clear and readable.arrow_forwardNew question pls answer (4, 5, & 6) only 1. Identify/name the specific molecule/monomer to the left. 2. Name the two/2 functional groups the molecule contains. 3. What specific molecule is formed when two of these molecules combine together? 4. What molecule is produced in humans when hundreds of these molecules combine together 5. Where in the human body would you find the largest concentration of the molecule mentioned in number 4 6. Name two polysaccharides that provide structural support to the organisms that contain them.arrow_forward
- Give complete iupac name of each. Draw the correct structure including carbons and hydrogens for 3&4arrow_forwarda.Identify the functional groups in the ball-and-stick model of neral, a compound with a lemony odor isolated from lemongrass. b. Draw a skeletal structure of a constitutional isomer of neral that should be more water soluble. c.Label the most electrophillic carbon atom.arrow_forwardWrite the IUPAC name for each of the following. (in condensed form if possible). * only solve a,b, & carrow_forward
- Answer each question using the ball-and-stick model of compound A. Give the IUPAC name for A, including R,S designations for stereogeniccenters.Classify A as a 1°, 2°, or 3° alcoholarrow_forwardQuestion from at-home practice: How many and what types of carbon atoms are present in your compound? (carbonyl, aromatic, alkene, etc.)arrow_forwardDraw the structural formula for each of the following:( In condensed formula and line angle) d. 3-propoxyheptanee. 1,3-dimethoxybenzenef. ethoxycyclobutanearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry In Focus
Chemistry
ISBN:9781305084476
Author:Tro, Nivaldo J., Neu, Don.
Publisher:Cengage Learning
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY