
Interpretation:
A concept map in which the effect of changes on system, reactants and products is shown according to Le-Chatelier’s principle is to be drawn.
Concept introduction:
According to Le-Chatelier’s principle, if a system at equilibrium experience any change then it responds in a way to cancel out that change.
Answer to Problem 17SSC
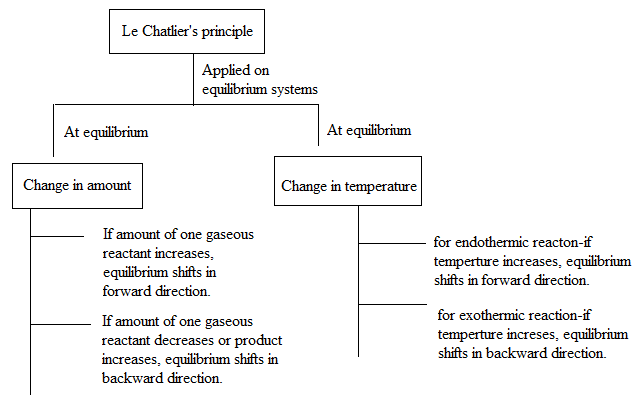
Explanation of Solution
The above diagram shows how addition of reactants or addition of products causes a shift in equilibrium in accordance to the Le-Chatelier’s principle.
- When there is any increase in concentration or pressure of reactants, equilibrium shifts in forward direction to cancel out the effect or to decrease the concentration or pressure of reactant.
- When there is any increase in concentration or pressure of products, equilibrium shifts in backward direction to cancel out the effect.
- At equilibrium, if increase in temperature takes place then the change depends on the type of the reaction.
For an endothermic reaction, increase in temperature shifts the equilibrium in forward direction. For an exothermic reaction, increase in temperature shifts the equilibrium in reverse direction.
Chapter 17 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
CHEMISTRY-TEXT
Chemistry: Structure and Properties
Organic Chemistry (8th Edition)
Essential Organic Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





