
Concept explainers
Calculate the pH after the addition of 35 mL of 0.10 M NaOH to 30 mL of 0.10 M HCN.
a)
11.89
b)
2.11
c)
12.22
d)
1.78
e)
1300
For questions 17.3.5 to 17.3.8, refer to the following diagrams. The solutions shown represent various points in the titration of the weak acid HA with NaOH. (For clarity, the sodium ions and water molecules are not shown.)
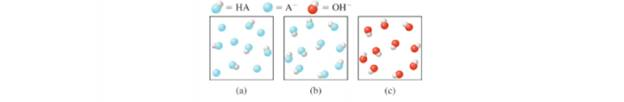
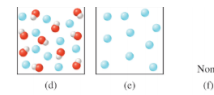
Interpretation:
The
Concept introduction:
Acid–base titration is a technique to analyze the unknown concentration of the acid or base with the help of the known concentration of the acid and base.
The equivalence point is the point in an acid–base titration in a chemical reaction where number of moles of the titrant and the unknown concentration of the analyte are equal. It is used to identify the unknown concentration of the analyte.
In strong acid–base titration, the pH of the solution is neutral (i.e., pH = 7) at equivalence point.
In weak acid–strong base titration, the pH of the solution is not neutral (pH> 7) at equivalence point.
In strong acid–weak base titration, the pH of the solution is not neutral (pH < 7) at equivalence point.
The number of moles of the molecule are calculated as:
The number of millimoles of a molecule are calculated as:
For acidic buffer, the value of
The
The relationship between
Answer to Problem 4CP
Solution: Option (a).
Explanation of Solution
Given information:
Theconcentration of
The equation for reaction between
This equation can be simplifiedas follows:
After addition of
Millimoles of the molecule are calculated using the expression as follows:
Millimoles of
Substitute the concentration of
Millimoles of
Substitute the concentration of
Summarise the millimoles at equilibrium as follows:
At equilibrium, the solution has astrongbase. The millimoles of
Total volume of the solution is
Concentration of
Substitute the millimoles of
The
Substitute the concentration of
The
Substitute the value of
The
Hence, the correct answer is option (a).
Reason for incorrect options:
Option (b) is incorrect because on solving with the help of the above equation, the answer does not match with option (b).
Option (c) is incorrect because on solving with the help of the above equation, the answer does not match with option (c).
Option (d) is incorrect because on solving with the help of the above equation, the answer does not match with option (d).
Option (e) is incorrect because on solving with the help of the above equation, the answer does not match with option (e).
Hence, options(b), (c), (d), and (e) are incorrect.
Want to see more full solutions like this?
Chapter 17 Solutions
Chemistry (Looseleaf) - With Student Solutions Manual
- A certain acetic acid solution has pH = 2.68. Calculate the volume of 0.0975 M KOH required to reach the equivalence point in the titration of 25.0 mL of the acetic acid solution.arrow_forwardA chemist needs a buffer with pH 3.50. How many milliliters of pure formic acid (density = 1.220 g/mL) must be added to 375 mL of 0.0857 M NaOH solution to obtain such a buffer?arrow_forwardA chemist needs a buffer with pH 4.35. How many milliliters of pure acetic acid (density = 1.049 g/mL) must be added to 465 mL of 0.0941 M NaOH solution to obtain such a buffer?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





