
(a)
Interpretation:
It should be determined that the product obtained from the reaction of Ethyl butanoate with
Concept introduction:
Carboxylic compounds react with
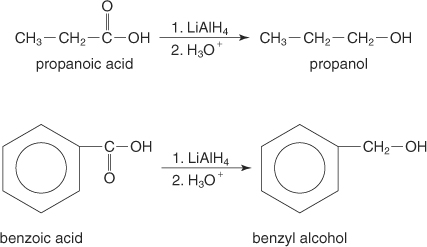
(b)
Interpretation:
It should be determined that the product obtained from the reaction of Benzoic acid with
Concept introduction:
Carboxylic compounds react with

(c)
Interpretation:
It should be determined that the product obtained from the reaction of Methyl benzoate with
Concept introduction:
Carboxylic compounds react with

(d)
Interpretation:
It should be determined that the product obtained from the reaction of Pentatonic acid
with
Concept introduction:
Carboxylic compounds react with

Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry (Looseleaf) - With Access
- Describe the Methods used to Synthesis of Acyl Chlorides or Acyl bromides.arrow_forwardThe oxidation of 3-methylbutanal with potassium permanganate in an acid medium produces compound A and the reduction of cyclohexanone with NaBH4 produces compound B. Indicate the CORRECT alternative: a) A reacts rapidly with water to produce a carboxylic acid. b) Reaction of B with methylamine produces an amide. c) Reaction of A with an acyl chloride produces an ester. d) B reacts with HCN to produce a cyanohydrin. e) The reaction of A and B in an acid medium produces cyclohexyl 3-methylbutanoate.arrow_forwardReaction of benzyl alcohol with ch3so2cl and pyridine and ch3ch2onaarrow_forward
- Esters and amides are most easily made by nucleophilic acyl substitution reactions on… A. alcohols B. acid chlorides C. acid anhydrides D. carboxylates E. carboxylic acidsarrow_forwardWhat is the reaction mechanism for formaldehyde and phenol under acidic conditions?arrow_forwardSYNTHESIS OF ESTERS VIA NUCLEOPHILIC ACYL SUBSTITUTION Write the chemical equation involved in the reaction between the excess acid and NaHCO3. Explain why NaHCO3 is preferred over NaOH for the neutralization of excess acid. How was excess alcohol eliminated from the crude product.arrow_forward
- What product is obtained from the reaction of excess benzene with isobutyl chloride + AlCl3 ?arrow_forwardWhat compounds are obtained when (+)-limonene reacts with O3 followed by dimethyl sulfide?arrow_forwardPhosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the following reagents? a. one equivalent of methanol b. excess methanol c. excess propylamine d. excess waterarrow_forward
- The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Propose a mechanism for the Ritter reaction.b. Why does the Ritter reaction not work with primary alcohols?c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forwardPropose a mechanism for the acid-catalyzed reaction of salicylic acid with aceticanhydride.(b) Explain why a single drop of sulfuric acid dramatically increases the reaction ratearrow_forwardwhat is the structure of o-methylphenoxide anion and what is its pKa?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





