
a) Styrene (PhCH=CH2)
Interpretation:
How to prepare styrene from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield
To state:
How to prepare styrene from 2-phenylethanol.
Answer to Problem 47AP
Styrene can be prepared by treating 2-phenylethanol with POCl3 in pyridine.
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene.
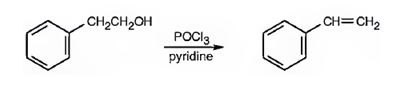
Styrene can be prepared by treating 2-phenylethanol with POCl3 in pyridine.
b) Phenylacetaldehyde (PhCH2CHO)
Interpretation:
How to prepare phenylacetaldehyde from 2-phenylethanol is to be stated.
Concept introduction:
Dess-Martin periodinate in dichloromethane oxidizes 10alcohols to
To state:
How to prepare phenylacetaldehyde from 2-phenylethanol
Answer to Problem 47AP
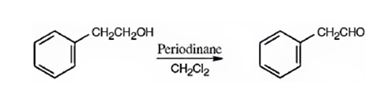
Phenylacetaldehyde can be prepared by oxidizing 2-phenylethanol with Dess-Martin periodinate in dichloromethane.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to phenylacetaldehyde when treated with Dess-Martin periodinate in dichloromethane.
Phenylacetaldehyde can be prepared by oxidizing 2-phenylethanol with Dess-Martin periodinate in dichloromethane.

c) Phenylacetic acid (PhCH2COOH)
Interpretation:
How to prepare phenylacetic acid from 2-phenylethanol is to be stated.
Concept introduction:
CrO3 in acidic solutions oxidize 10 alcohols directly into acids and 20 alcohols to ketones. It does not oxidize 30 alcohols.
To state:
How to prepare phenylacetic acid from 2-phenylethanol.
Answer to Problem 47AP
Phenylacetic acid can be prepared by oxidizing 2-phenylethanol with CrO3 in acidic solutions.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to phenylacetic acid when treated with CrO3 in acidic solutions.
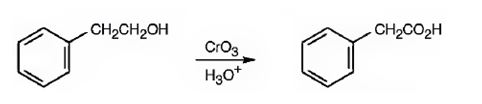
Phenylacetic acid can be prepared by oxidizing 2-phenylethanol with CrO3 in acidic solutions.
d) Benzoic acid
Interpretation:
How to prepare benzoic acid from 2-phenylethanol is to be stated.
Concept introduction:
KMnO4 in acidic solutions oxidize
To state:
How to prepare benzoic acid from 2-phenylethanol.
Answer to Problem 47AP
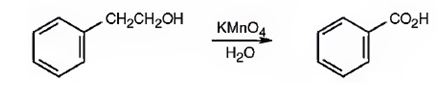
Benzoic acid can be prepared by oxidizing 2-phenylethanol with KMnO4 in acidic solutions.
Explanation of Solution
2-phenylethanol is a 10 alcohol. It gets oxidized to benzoic acid when treated with KMnO4 in acidic solutions.
Benzoic acid can be prepared by oxidizing 2-phenylethanol with KMnO4 in acidic solutions.
e) Ethylbenzene
Interpretation:
How to prepare ethylbenzene from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene upon reduction gives the
To state:
How to prepare ethylbenzene from 2-phenylethanol.
Answer to Problem 47AP
Ethylbenzene can be prepared from 2-phenylethanol by following the steps shown below.
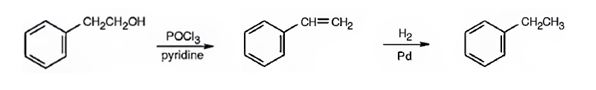
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When treated with H2/Pd, the double bond in the side chain gets reduced to yield ethyl benzene.
Ethylbenzene can be prepared from 2-phenylethanol by following the steps shown below.

f) benzaldehyde
Interpretation:
How to prepare benzaldehyde from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene upon ozonolyzis followed by reduction will yield the aldehyde required.
To state:
How to prepare benzaldehyde from 2-phenylethanol.
Answer to Problem 47AP
Benzaldehyde can be prepared from 2-phenylethanol by following the steps shown below.
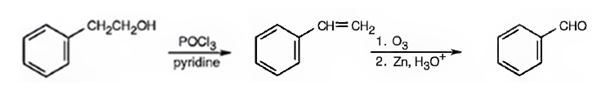
Explanation of Solution
2-Phenylethanol when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When styrene is subjected to ozonolysis followed by reduction, the double bond in side chain gets cleaved resulting in the formation of benzaldehyde.
Benzaldehyde can be prepared from 2-phenylethanol by following the steps shown below.

g) 1-phenylethanol
Interpretation:
How to prepare 1-phenylethanol from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols undergo dehydration when treated with POCl3 in pyridine to yield an alkene. The alkene adds a molecule of water following oxymercuration-demercuration process. The addition will take place following Markovnikov regiochemistry.
To state:
How to prepare 1-phenylethanol from 2-phenylethanol.
Answer to Problem 47AP
1- Phenylethanol can be prepared from 2-phenylethanol by following the steps shown below.
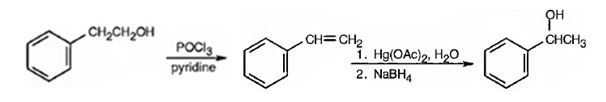
Explanation of Solution
2-Phenylethanol, when treated with POCl3 in pyridine eliminates a molecule of water from the side chain to yield styrene. When styrene is subjected oxymercuration-demercuration processes, a molecule of water is added, following Markovnikov regiochemistry, to the double bond. The –OH adds on to the more alkyl substituted carbon and H to the less alkyl substituted carbon in double bond to yield 1-phenylethanol.
1-Phenylethanol can be prepared from 2-phenylethanol by following the steps shown below.

h) 1-Bromo-2-phenylethane
Interpretation:
How to prepare 1-bromo-2-phenylethane from 2-phenylethanol is to be stated.
Concept introduction:
Alcohols yield the corresponding alkyl bromides when treated with PBr3.
To state:
How to prepare 1-bromo-2-phenylethane from 2-phenylethanol is to be stated.
Answer to Problem 47AP
1-Bromo-2-phenylethane can be prepared from 2-phenylethanol by following the steps shown below.
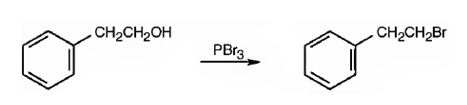
Explanation of Solution
When 2-phenylethanol is treated with PBr3, a bimolecular nucleophilic substitution of –OH by Br takes place to yield 1-bromo-2-phenylethane.
1-Bromo-2-phenylethane can be prepared from 2-phenylethanol by following the steps shown below.

Want to see more full solutions like this?
Chapter 17 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(a) PhMgBr, then H3O+ (b) Tollens reagent (c) semicarbazide and weak acid(d) excess ethanol and acid (e) propane-1,3-diol, H+ (f) zinc amalgam and dilute hydrochloric acidarrow_forwardThe following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardHow could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) Pentanoic acid (d) 1-Butene (e) Octanearrow_forward
- Give reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forwardGive reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forward
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with the following reagents.(a) isobutylene and HF (b) cyclohexanol and BF3 (c) fuming sulfuric acidarrow_forwardCompound X (C4H9Br) reacts by heating with NaOH in H2O to form Y. The compound Y then undergoes acid catalysed hydration by H2SO4 in 180°C to form 2-methyl prop-1-ene. (e) Determine the structure of X and Y. (f) Predict a MAJOR product when compound Y reacts with H2SO4 in 140°C. (g) Draw a structural isomer of X. Name the isomer using IUPAC nomenclature. (h) Describe a chemical test to distinguish between compound Y and 1-butanol.arrow_forwardHow would you prepare the following compounds from 2-phenylethanol? More than one step may be required. (a) Styrene (PHCH=CH2) (b) Phenylacetaldehyde (PHCH2CHO) (c) Phenylacetic acid (PHCH2COOH) (d) Benzoic acid (e) Ethylbenzene (f) Benzaldehyde (g) 1 -Phenylethanol (h) l-Bromo-2-phenylethanearrow_forward
- Predict which member of each pair is more acidic, and explain the reasons for your predictions.(a) cyclopentanol or 3-chlorophenol (b) cyclohexanol or cyclohexanethiol(c) cyclohexanol or cyclohexanecarboxylic acid (d) butan-1-ol or 2,2-dichlorobutan-1-olarrow_forward16-57 Starting with either benzene or toluene, how would you synthesize the following substances? Assume that ortho and para isomers can be separated. (a) 2-Bromo-4-nitrotoluene (c) 2,4,6-Tribromoaniline (b) 1,3,5-Trinitrobenzene (d) m-Fluorobenzoic acidarrow_forward3.) Starting from phenol, the synthesis of 2,4-dichlorophenoxyacetic acid involves four steps outlined by the roadmap below. Please provide the missing reagents and products where necessary. OH 1. NaOH HCl(aq) 2 Cl₂ FeCla OH 2,4-dichlorophenoxyacetic acid Iarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





