
Concept explainers
Interpretation:
A structure for the alcohol with molecular formula C9H12O and the following spectral data is to be proposed.
1HNMR: 0.88 δ (triplet,Rel.area=3.00); 1.80 δ (quintet, Rel.area=2.00); 2.32 δ (Rel.area=1.00); 4.54 δ (triplet, Rel.area=1.00); 7.24 δ (Rel.area=5.00).
Concept introduction:
In 1HNMR spectrum, the alcoholic proton absorption occurs in the range 3.4 δ - 4.5 δ while that of phenolic proton occur in the range 3.0 to 8.0. The
The absorption due to 10 alkyl group (CH3) is seen around 0.7 δ - 1.3 δ, that due to a 20 alkyl group (CH2) is seen around 1.2 δ - 1.6 δ while that due to 30 alkyl group (CH) is seen around 1.4 δ - 1.8 δ The multiplicity of a signal gives an idea about the protons present in the adjacent carbons.
To propose:
A structure for the alcohol with molecular formula C9H12O and the following spectral data is to be proposed.
1HNMR: 0.88 δ (triplet,Rel.area=3.00); 1.80 δ (quintet, Rel.area=2.00); 2.32 δ (Rel.area=1.00); 4.54 δ (triplet, Rel.area=1.00); 7.24 δ (Rel.area=5.00).
b)
C8H10O2
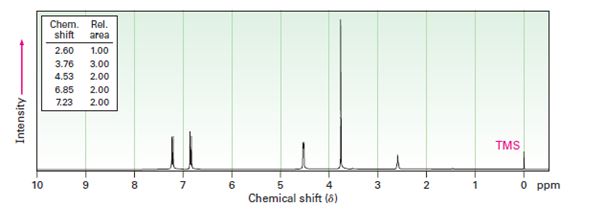
Interpretation:
A structure for the alcohol with molecular formula C8H10O2 and the following spectral data is to be proposed.
IR: 3500 cm-1,
1HNMR: 2.60 δ (Rel.area=1.00); 3.76 δ (Rel.area=3.00); 4.53 δ (Rel.area=2.00); 6.85 δ (Rel.area=2.00); 7.23 δ (Rel.area=2.00).
Concept introduction:
In 1HNMR spectrum, the alcoholic proton absorption occurs in the range 3.4 δ - 4.5 δ while that of phenolic proton occur in the range 3.0 to 8.0. The aromatic protons give a broad peak in the range 6.5 δ - 8.0 δ, the benzylic protons normally absorb in the region 2.3 δ - 3.0 δ.
To propose:
A structure for the alcohol with molecular formula C8H10O2 and the following spectral data is to be proposed.
1HNMR: 2.60 δ (Rel.area=1.00); 3.76 δ (Rel.area=3.00); 4.53 δ (Rel.area=2.00); 6.85 δ (Rel.area=2.00); 7.23 δ (Rel.area=2.00).
Trending nowThis is a popular solution!

Chapter 17 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- The 1H and 13C NMR spectra below belong to a compound with formula C6H10O2. Propose a structure for this compound.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardThe following sequence of steps converts (R)-2-octanol to (S)-2-octanol. Propose structural formulas for intermediates A and B, specify the configuration of each, and account for the inversion of configuration in this sequence.arrow_forward
- Cyclohexene can be converted to 1-cyclopentenecarbaldehyde by the following series of reactions. Propose a structural formula for each intermediate compound.arrow_forwardAmines are converted into alkenes by a two-step process called Hofmann elimination. SN2 reaction of the amine with an excess of CH3I in the first step yields an intermediate that undergoes E2 reaction when treated with silver oxide as base. Pentylamine, for example, yields 1-pentene. Propose a structure for the intermediate, and explain why it readily undergoes elimination.arrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forward
- Treatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products(M and N) with molecular formula C6H12. The 1H NMR spectra of M and Nare given below. Propose structures for M and N, and draw a mechanismto explain their formation.arrow_forwardPredict the major products of the following reactions, and propose mechanisms tosupport your predictions.(a) pent-1-ene + HClarrow_forwardA compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forward
- Hydroboration of 2-methyl-2-pentene at 25°C, followed by oxidation with alkaline H2o2, yields 2-methyl-3-pentanol, but hydroboration at 160°C followed by oxidation yields 4-methyl-1-pentanol. Suggest a mechanism.arrow_forwardCompounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm−1 . The 1H NMR spectra of Y and Z are given below. Propose structures for Y and Z.arrow_forwardTreatment of trans-2-chlorocyclohexanol with NaOH yields 1, 2-epoxy-cyclohexane, but reaction of the cis isomer under the same conditions yields cyclohexanone. Propose mechanisms for both reactions, and explain why the different results are obtained.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

