
ORGANIC CHEMISTRY-ETEXT REG ACCESS
12th Edition
ISBN: 9781119308362
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 16P
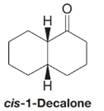
Treating a solution of cis-1-decalone with base causes an isomerization to take place. When the system reaches equilibrium, the solution is found to contain about 95% trans-1-decalone and about 5% cis-1-decalone. Explain this isomerization.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Organic Chemistry
Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major product. Also acid catalyzed dehydration reaction of 3-methyl-1-butanol give the same product as major product. Explain the reason why both of the reaction produce the same product as the major product.
When trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone.
Why doesn’t the cis isomer yield the oxide?
Write down the common (not IUPAC) names of the organic molecules that would be released if this molecule were hydrolyzed:
CH2−O−C—(CH2);–CH=CH–CH2–CH=CH—(CH2)4—CH3
CH-O-C-(CH2)14-CH3
O
11
CH2−O−C— (CH2)14 — CH3
Separate each name with a comma. You will find useful information in the ALEKS Data resource.
1
010
Continue
O
a
X
000
Y
F8
F9
Submi
Chapter 18 Solutions
ORGANIC CHEMISTRY-ETEXT REG ACCESS
Ch. 18 - Prob. 1PPCh. 18 - Practice Problem 18.2 Would optically active...Ch. 18 - Prob. 3PPCh. 18 - Practice Problem 18.4 Why do we say that the...Ch. 18 - Prob. 5PPCh. 18 - Practice Problem 18.6 (a) Write a reaction...Ch. 18 - PRACTICE PROBLEM 18.7
Show how you would use the...Ch. 18 - Practice Problem 18.8 The acetoacetic ester...Ch. 18 - Practice Problem 18.9
In the synthesis of the keto...Ch. 18 - PRACTICE PROBLEM 18.10 How would you use the...
Ch. 18 - PRACTICE PROBLEM 18.11
How would you use the...Ch. 18 - PRACTICE PROBLEM 18.12 Outline all steps in a...Ch. 18 - PRACTICE PROBLEM 18.13
The antiepileptic drug...Ch. 18 - PRACTICE PROBLEM 18.14 Show how you could employ...Ch. 18 - Prob. 15PCh. 18 - Treating a solution of cis-1-decalone with base...Ch. 18 - Prob. 17PCh. 18 - Prob. 18PCh. 18 - Prob. 19PCh. 18 - Prob. 20PCh. 18 - Prob. 21PCh. 18 - Prob. 22PCh. 18 - Prob. 23PCh. 18 - The synthesis of cyclobutanecarboxylic acid given...Ch. 18 - Prob. 25PCh. 18 - Prob. 26PCh. 18 - Prob. 27PCh. 18 - Prob. 28PCh. 18 - Compound J, a compound with two four-membered...Ch. 18 - Prob. 30PCh. 18 - Prob. 31PCh. 18 - 18.32 Shown below is a synthesis of the elm bark...Ch. 18 - 18.33 (a) A compound U gives a negative iodoform...Ch. 18 - 18.34 Compound A has the molecular formula and...Ch. 18 - Prob. 35PCh. 18 - Prob. 36PCh. 18 - Prob. 37PCh. 18 - Prob. 38PCh. 18 - 1. -Carotene is a highly conjugated hydrocarbon...Ch. 18 - Dehydroabietic acid is a natural product isolated...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/z value of 128.
Organic Chemistry (8th Edition)
1.1 Write a one-sentence definition for each of the following:
a. chemistry
b. chemical
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
The percent composition of the elements in a molecule of cholesterol ( C27H45OH ) needs to be determined. Conce...
Chemistry: Matter and Change
One of the following statements is true and the other as false regarding the first-order reaction 2AB+C . Ident...
General Chemistry: Principles and Modern Applications (11th Edition)
The reaction of tert-butyl chloride with methanol (CH3)3CCl + CH3OH (CH3)3COCH3 + HCl tert butyl chloride met...
Organic Chemistry (9th Edition)
39. What are the units of k for each type of reaction?
a. first-order reaction
b. second-order reaction
c...
Chemistry: Structure and Properties
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Acid-catalyzed dehydration of 3-methyl-2-pentanol gives three alkenes: 3-methyl-1-pentene, 3-methyl-2-pentene, and 3-methylenepentane. Draw the structure of the carbocation intermediate leading to the formation of 3-methyl-2-pentene.arrow_forwardWhat is the IUPAC name of this compound? O 2-Ethyl-4-methyl-1,5-hexadione O 5-Ethyl-5-formyl-3-methyl-2-heptanone O2-Ethyl-4-methyl-5-oxo-2-hexanone O2-Ethyl-4-methyl-5-oxohexanal O2-Ethyl-4-methyl-2,6-hexadialarrow_forward1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-octen-3-ol into a less effective molecule. Draw the structure of a hydrogenation product of 1-octen-3-ol.arrow_forward
- THC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THCarrow_forwardWhy is the boiling point of propane-1,3-diol (HOCH2CH2CH2OH) higher than the boiling point of propane-1,2-diol [HOCH2CH(OH)CH3] (215 °C vs. 187 °C)? Why do both diols have a higher boiling point than butan-1-ol (CH3CH2CH2CH2OH, 118 °C)?arrow_forwardWhat is the IUPAC name of this compound? H 5-Ethyl-5-formyl-3-methyl-2-heptanone 2-Ethyl-4-methyl-5-oxohexanal 2-Ethyl-4-methyl-2,6-hexadial 2-Ethyl-4-methyl-5-oxo-2-hexanone 2-Ethyl-4-methyl-1,5-hexadionearrow_forward
- Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major product. Also acid catalyzed dehydration reaction of 3-methyl-1-butanol give the same product as major product. Explain the reason why both of the reaction produce the same product as the major product.arrow_forwardWrite in the correct reagents for these transformations: a) cyclohexanol -→ chlorocyclohexane b) 2-butanone 2-butanol ward 2-butanol 2-butanone toluene - → benzoic acid nitrobenzene → anilinearrow_forwardWhich of the following reactions is a substitution reaction? C6H6 + Cl2 → C6H5Cl + HCl CH2=CH2 + Cl2 → CH2ClCH2Cl CH2=CH2 + HCl → CH3CH2Cl CH2=CH2 + H2 → CH3CH3 HCCH + 2Cl2 → CHCl2CHCl2arrow_forward
- Alkenes can be converted to alcohols by reaction with mercuric acetate to form a ß-hydroxyalkylmercury(II) acetate compound, a reaction called oxymercuration. Subsequent reduction with NaBH reduces the C-Hg bond to a C-H bond, forming the alkyl alcohol, a reaction called demercuration. Draw the structure of the Hg-containing compound and the final alcohol product formed in the reaction sequence. CH3 Hg(OOCCH3)2 NaBH4 HO™ Oxymercuration product Demercuration Product H₂O, THF Draw the alcohol product of demercuration. Draw the neutral product of oxymercuration. Omit byproducts. OH Hg Incorrectarrow_forwardChoose the best explanation for why these compounds are all less than fifteen carbons. A) Aldehydes with longer alkyl chains become increasingly nonpolar and the LDF are stronger than hydrogen bonding with ethanol. B) Ethanol molecules will hydrogen bond with the oxygen of the aldehydes. However, longer alkyl chains interrupt the ability of the aldehyde to hydrogen bond with ethanol which decreases the solubility. C) Ethanol molecules will form strong LDF interactions between the alkyl chains of the aldehydes and the alkyl groups of ethanol. The strong attractions cause the aldehydes to precipitate out of solution. D) When aldehydes chain length increases, the aldehydes have stronger intermolecular force with the small ethanol solvent molecules than the ethanol-ethanol intermolecular force.arrow_forwardName this aldehyde/ketone according to the IUPAC system. Include stereochemistry. CH3OCH₂CH2 H CH₂CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY