
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 18.41P
You have learned two ways to make an alkyl benzene: Friedel–Crafts alkylation, and Friedel–Crafts acylation followed by reduction. Although some alkyl benzenes can be prepared by both methods, it is often true that only one method can be used to prepare a given alkyl benzene. Which method(s) can be used to prepare each of the following compounds from benzene? Show the steps that would be used.
a. ![]() b.
b. 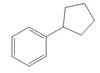 c.
c. 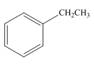 d.
d. 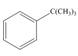
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
You have learned two ways to make an alkyl benzene: Friedel–Craftsalkylation, and Friedel–Crafts acylation followed by reduction. Although some alkyl benzenes can be prepared by both methods, it is often true that only one method can be used to prepare a given alkyl benzene. Which method(s) can be used to prepare attached compound from benzene? Show the steps that would be used.
What is the best way to turn benzene into the following product? Would friedel-crafts alkylation followed by some sort of nitration work or would that be too low yield?
Why does the carbomethoxy group directs the reaction to positions that are meta to it ?b) Why the formation of dinitrobenzaote is substantially disfavored ?c) Would you expect small amounts of the ortho and para substituted product ? How would you
remove them if they are formed ?d) Why does water have a retarding effect on the nitration ?e) Explain why Benzene has lower reactivity in electrophilic addition reactions than cyclohexene?
Chapter 18 Solutions
Organic Chemistry
Ch. 18 - Prob. 18.1PCh. 18 - Prob. 18.2PCh. 18 - Prob. 18.3PCh. 18 - Prob. 18.4PCh. 18 - Prob. 18.5PCh. 18 - What acid chloride would be needed to prepare each...Ch. 18 - Prob. 18.7PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.9PCh. 18 - Prob. 18.10P
Ch. 18 - Prob. 18.11PCh. 18 - Prob. 18.12PCh. 18 - Problem 18.14 Draw all resonance structures for...Ch. 18 - Classify each substituent as electron donating or...Ch. 18 - Prob. 18.15PCh. 18 - Label each compound as more or less reactive than...Ch. 18 - Rank the following compounds in order of...Ch. 18 - Problem 18.18 Rank the following compounds in...Ch. 18 - Prob. 18.19PCh. 18 - Problem 18.20 Draw the products of each...Ch. 18 - Prob. 18.21PCh. 18 - Prob. 18.22PCh. 18 - Devise a synthesis of each compound from the...Ch. 18 - Problem 18.24 Draw the products of each...Ch. 18 - Problem 18.25 Draw a stepwise mechanism for the...Ch. 18 - Problem 18.26 Draw the products of each...Ch. 18 - Prob. 18.27PCh. 18 - Prob. 18.28PCh. 18 - Problem 18.29 How could you use ethylbenzene to...Ch. 18 - Prob. 18.30PCh. 18 - Problem 18.31 What steps are needed to convert...Ch. 18 - Problem 18.32 Synthesize each compound from...Ch. 18 - Synthesize each compound from benzene.Ch. 18 - Prob. 18.34PCh. 18 - 18.35 What is the major product formed by an...Ch. 18 - Draw the products formed when phenol (C6H5OH) is...Ch. 18 - Prob. 18.37PCh. 18 - Draw the products of each reaction. a. e. i. b. f....Ch. 18 - What products are formed when benzene is treated...Ch. 18 - Draw the products of each reaction. a.d. b.e. c.f.Ch. 18 - You have learned two ways to make an alkyl...Ch. 18 - Prob. 18.42PCh. 18 - Prob. 18.43PCh. 18 - 18.45 Explain why each of the following reactions...Ch. 18 - Prob. 18.45PCh. 18 - 18.47 For each of the following substituted...Ch. 18 - Which benzene ring in each compound is more...Ch. 18 - 18.49 For each N-substituted benzene, predict...Ch. 18 - Explain each statement in detail using resonance...Ch. 18 - Prob. 18.50PCh. 18 - Explain why the meta product is formed in the...Ch. 18 - 18.53 Rank the aryl halides in each group in order...Ch. 18 - Prob. 18.53PCh. 18 - 18.54 Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.55PCh. 18 - 18.56 Draw a stepwise, detailed mechanism for the...Ch. 18 - Prob. 18.57PCh. 18 - 18.58 Draw a stepwise mechanism for the following...Ch. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.60PCh. 18 - Draw a stepwise mechanism for the following...Ch. 18 - Prob. 18.62PCh. 18 - Prob. 18.63PCh. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from benzene and any...Ch. 18 - Synthesize each compound from toluene (C6H5CH3)...Ch. 18 - Prob. 18.67PCh. 18 - Use the reactions in this chapter along with those...Ch. 18 - Prob. 18.69PCh. 18 - Prob. 18.70PCh. 18 - Problem 18.69 Identify the structures of isomers A...Ch. 18 - Prob. 18.72PCh. 18 - Problem 18.71 Compound X (molecular formula ) was...Ch. 18 - Prob. 18.74PCh. 18 - The NMR spectrum of phenol () shows three...Ch. 18 - Explain the reactivity and orientation effects...Ch. 18 - Prob. 18.77PCh. 18 - Prob. 18.78PCh. 18 - Prob. 18.79PCh. 18 - Prob. 18.80P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the Friedel-Crafts alkylation of benzene, dialkylation is often a significant by-product. In the Friedel-Crafts acylation of benzene, diacylation is not a significant by-product. Which of the following is the primary reason for this difference? a. Acyl cations are more difficult to make with Lewis acids b.Unlike acyl cations, carbocations can undergo rearrangements. c.Alkyl groups activate the ring to further substitution, acyl groups deactivate it. d. Alkyl groups activate the ring to further substitution, acyl groups deactivate itarrow_forwardWhat two sets of reagents (each consisting of a carbonyl compound and phosphonium ylide) can be used for the synthesis of the following alkene? a. What alkyl halide is required to prepare each of the phosphonium ylides? b. What is the best set of reagents to use for the synthesis?arrow_forwardInternal alkynes are able to selectively form one product when subjected to most reaction conditions. T or F Alkynes react with one equivalent of acid to create a carbocation for Markovnikov addition. T or F Alkynes react very similarly to alkenes. Understanding the mechanisms from chapter 12 will help understand the reaction pathways in chapter 13 where mechanisms are not explicitly drawn. T or Farrow_forward
- The formation of biphenyl from benzene ring as shown below takes place using Friedel-Crafts alkylation. TRUE OR FALSEarrow_forwardRank the alkyl halides in the following marine natural product in order of increasing reactivity in the SN2 reaction.arrow_forwardThe following compound readily eliminates CO; to form a conjugated six membered ring. (a) Complete the reaction with drawing the possible structure and (b) explain why this reaction goes steadily.arrow_forward
- Which product would be obtained if 3-methyl-2-butanol was used as the alcohol in the Electrophilic Aromatic Substitution: Friedel-Crafts Alkylation? Show the structure of the product. Explain briefly. Original reaction and more info is attached:arrow_forwardGive the major organic product of the following reaction sequences (d & e).arrow_forwardA student in Ochem proposed the following synthesis. Explain the problem with this approach and provide the necessary reagents to convert halohydrin A to alkene B (more than one step is required) in addition provide the product after each step. Provide the mechanism and expected actual final product from the reaction of halohydrin A with LDAarrow_forward
- The anticoagulant Warfarin can be synthesized by a conjugate addition (Michael addition) reaction as shown below: a) Propose a mechanism for the reaction starting from the enolate shown below. You can assume water is present for any proton transfers needed.b) We saw that the keto tautomer is normally preferred over the enol tautomer, however, the enol tautomer is present in the final product (Warfarin) and that is the more stable tautomer in this case. Why do you think the enol tautomer is more stable? Hint: consider the molecule as a whole.arrow_forwardIn the Friedel Crafts Alkylation of benzene with 2-chloro-1-phenylpropane, the expected product is not produced. Instead of 1,2-diphenylpropane (expected product), the product 1,1-diphenylpropane (observed product) is produced (see below). Explain why this product is produced.arrow_forwardConsidering the mechanism for Friedel-Crafts alkylation, how would you explain the formation of this particular product?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License