
Concept explainers
For each of the following substituted benzenes: [1]
a. Does the substituent donate or withdraw electron density by an inductive effect?
b. Does the substituent donate or withdraw electron density by a resonance effect?
c. On balance, does the substituent make a benzene ring more or less electron rich than benzene itself?
d. Does the substituent activate or deactivate the benzene ring in electrophilic
(a)
Interpretation: The substituents present on the given benzene donate or withdraw electron density by an inductive effect is to be predicted.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.47P
The substituent in
Explanation of Solution
The inductive effect appears in a molecule due to the difference in electron negativity of atoms. The groups attached to benzene ring in the given compound are
The electron negativity of bromine is more than carbon atom. Hence, the substituent in
The electron negativity of nitrogen is more than carbon atom. Hence, the substituent in
The electron negativity of oxygen is more than carbon atom. Hence, the substituent in
The substituent in
(b)
Interpretation: The substituents present on the given benzene donate or withdraw electron density by a resonance effect is to be predicted.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.47P
The substituent in
Explanation of Solution
The positive resonance effect is observed when the substituent present on a benzene ring donates its lone pair of electrons. Whereas, the negative resonance effect is observed, when the substituents present on a benzene ring pull its electrons towards itself.
The bromine atom attached to a benzene ring in
The carbon atom of
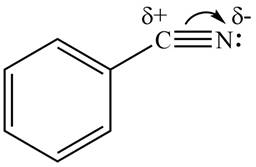
Figure 1
Therefore, the substituent in
The oxygen atom attached to a benzene ring in
The substituent in
(c)
Interpretation: The substituents make benzene ring more or less electron than benzene itself is to be predicted.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.47P
Substituents
Explanation of Solution
The electron donating groups or activating groups make benzene ring more electron rich than benzene itself, whereas the electron withdrawing groups or deactivating groups make benzene ring less electron rich than benzene itself.
The
Substituents
(d)
Interpretation: The substituents activate or deactivate the benzene ring in electrophilic aromatic substitution is to be predicted.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.47P
Substituents
Explanation of Solution
The electron donating groups activate the benzene ring in electrophilic aromatic substitution, whereas the electron withdrawing groups deactivate the benzene ring in electrophilic aromatic substitution.
The
Substituents
Want to see more full solutions like this?
Chapter 18 Solutions
ORGANIC CHEMISTRY
- 3 E C C Essentials of General, Organic, and Biochemistry Denise Guinn THIRD EDITION Draw a skeletal line structure for the aldehydes with the following condensed notations. 27 CH,CH,CH,CHO $ 4 R F V 5 Search or type URL T G stv♫ B 6 MacBook Pro Y H N & 7 U J 00 * 8 M TIZAC O I ✪ CH,(CH,),CHO K ( 9 < O L command ) O ◄ P presented by Macmillan Learning L option { باب ? 1 + 11 = AND Am } ] delete return shiftarrow_forward22:22 Tue 11 Jan * 84% Done Assignment 3 (3 of 9) 1. Identify the functional groups in the following molecules: (a) (b) (c) H. H С—ОН С-ОН Н-С—ОН C=C H H HO-C-H C-CH3 Н-С—ОН Acrylic acid (2 functional groups) H-C-OH Aspirin (3 functional groups) CH2OH Glucose (6 functional groups) 2. Identify the carbon atoms in the following molecules as primary, secondary tertiary, or quaternary: (a) CH3 (b) CH3CHCH3 (c) CH3 CH3 CH3CHCH2CH2CH3 CH3CH2CHCH2CH3 CH3CHCH,CCH3 ČH3 3. Give IUPAC names for the following alkanes: (a) CH3 (b) CH3 CH3CHCH2CH2CH3 CH3CH2ĊCH3 ČH3 (c) H3C CH3 (d) CH2CH3 CH3 CH3CHCCH2CH2CH3 CH3CH2CHCH2CH2CHCH3 CH3 (e) CH3 CH2CH3 (f) H3C CH3 CH3CH2CH2CHCH2CCH3 CH3C-CCH2CH2CH3 CH3 H3C CH3 4. Draw structures for the following substances: (a) 2-Methylheptane (c) 4-Ethyl-3,4-dimethyloctane (e) 1,1-Dimethylcyclopentane (b) 4-Ethyl-2-methylhexane (d) 2,4,4-Trimethylheptane (f) 4-Isopropyl-3-methylheptanearrow_forwardDraw the structural formula for the product formed upon hydroboration/oxidation of the Alkene below.arrow_forward
- Why is benzene less reactive toward electrophiles than an alkene, even though it has more π electrons than an alkene (six versus two)?arrow_forwardLabel the R group(s) in each of the following compounds. CH;-CH,-CH,-0–CH,–CH,–CH3 a. b. CH3-CH,-CH,-OH CH;-CH,-C-OH с.arrow_forwarddraw the structure(s) of the major organic product(s)arrow_forward
- What is true about the relationship between the following compounds? CH3 CH3 CH; CH; CH3 CH3 A В O All three are stereoisomers of each other. OA and B are the same compound while C is a stereoisomer. All three are the same compound. A and B are the same while C is a conformer, all of the same compound . O A and C are the same while B is a stereoisomer.arrow_forwardFor the substituted cyclohexane compound shown, identify the atoms that are trans to the bromo substituentarrow_forward35 Q1: 3. Discuss carbenes, in particular preparation of both singlet and triplet carbine and stereoselectivity in their reaction with cis-but-2-ene cis-but-2-ene إجابتكarrow_forward
- Draw a structural formula for the major organic product of the following reaction: CH₂Cl₂ Br₂ Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. . Do not show stereochemistry in other cases. If enantiomers are formed, just draw one. - 000 - IFarrow_forwardDraw a structural formula for the product formed upon hydroboration/oxidation of the alkene below. CH3CH2 C=CH2 CH3CH2 Use wedge and hash bonds ONLY for rings. Do not show stereochemistry in other cases. • If the reaction produces a racemic mixture, just draw one stereoisomer. C P opy aste ○, [片arrow_forwardThe chiral catalyst (R)-BINAP-Ru is used to hydrogenate alkenes to give alkanes . The products are produced with high enantiomeric excess. An example is the formation of (S)-naproxen, a pain reliever. Q.How can one enantiomer of naproxen be formed in such high yield?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
