
Concept explainers
The organic anion
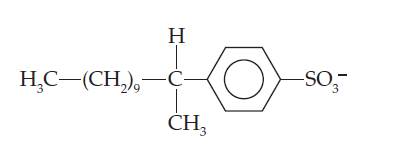
is found in most detergents. Assume that the anion undergoes aerobic decomposition in the following manner:
What is the total mass of O2 required to biodegrade 10.0 g of this substance?
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Laboratory Experiments for Chemistry: The Central Science (13th Edition)
- 7-43 (Chemical Connections 7A and 7B) Why is a high fever dangerous? Why is a low body temperature dangerous?arrow_forward7. Sulfuryl chloride, SO2Cl2, is a highly reactive gaseous compound. When heated, it decomposes as follows: SO2Cl2 (g) ⇌ SO2 (g) + Cl2 (g). This decomposition is endothermic. A sample of 3.509 grams of SO2Cl2 is placed in an evacuated 1.00-L bulb and the temperature is raised to 375 K. a. What would be the pressure (in atm) in the bulb if no dissociation of the SO2Cl2(g) occurred? b. When the system has come to equilibrium at 375 K, the total pressure in the bulb is found to be 1.43 atm. Calculate the partial pressures of SO2, Cl2, and SO2Cl2 at equilibrium. c. Give the expression for the equilibrium constant (either Kp or KC) for the decomposition of SO2Cl2(g) at 375 K. Calculate the value of the equilibrium constant you have selected. d. If the temperature were raised to 500 K, what effect would this have on the equilibrium constant? Explain in 1-2 sentences.arrow_forwardconsider the reaction 2A + 2B + C -›D + 3E A plot of Ln (pressure in torr) versus 1/ T (in kelvin) produce a straight line with an equation of the line Y=-23.70X+ 1.56 Calculate the pressure (in torr) of this reaction if the reaction is carried at 25.0degree Carrow_forward
- what is the equation for Delta E in chemistry?arrow_forwardThe organic anion is found in most detergents. Assume that the anion undergoesaerobic decomposition in the following manner: What is the total mass of O2 required to biodegrade 10.0 gof this substance?arrow_forwardIDENTIFY the following types of cheimcal reaction: S + O2 = SO2arrow_forward
- Which term refers to the movement of molecules from an area of higher concentration to an area of lower concentration?Single choice. A. Collision B. diffusion C. active transportarrow_forwardhow to use the equation of the line to calculate the arrhenius pre-exponential factor a y = -1.3376 x 10^4x + 34.073arrow_forwardThe decomposition of ozone in the stratosphere can occur by the following two-step mechanism: Br + O 3 → BrO + O 2 BrO + O → Br + O 2Which species is a catalyst in this mechanism? BrO O O3 Brarrow_forward
- The following reversible enzymatic reaction occurs in many areas of the body, but especially in red blood cells. The conversion of water and carbon dioxide into bicarbonate and hydrogen ions occurs through the action of an enzyme called carbonic anhydrase. Use the Law of Mass Action to predict which direction this equation will go under different circumstances. CO2 + H2O ↔ H+ + HCO3- Hyperventilating would increase the loss of carbon dioxide (CO2) from the blood. In what direction would the reaction above go in order to restore equilibrium? Left or right?arrow_forwardAn atmospheric chemist fills a container with gaseous N₂O₅ to a pressure of 125 kPa, and the gas decomposes to O₂. What is the partial pressure of NO₂, (in kPa),when the total pressure is 178 kPa?arrow_forwardOne of the many remarkable enzymes in the human bodyis carbonic anhydrase, which catalyzes the interconversionof carbon dioxide and water with bicarbonate ionand protons. If it were not for this enzyme, the body couldnot rid itself rapidly enough of the CO2 accumulated bycell metabolism. The enzyme catalyzes the dehydration(release to air) of up to 107 CO2 molecules per second.Which components of this description correspond to theterms enzyme, substrate, and turnover number?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





