
Concept explainers
GO Icicles. Liquid water coats an active (growing) icicle and extends up a short, narrow tube along the central axis (Fig 18-55). Because the water–ice interface must have a temperature of 0°C, the water in the tube cannot lose energy through the sides of the icicle or down through the tip because there is no temperature change in those directions. It can lose energy and freeze only by sending energy up (through distance L) to the top of the icicle, where the temperature Tr can be below 0°C. Take L = 0.12 m and Tr = −5°C. Assume that the central tube and the upward
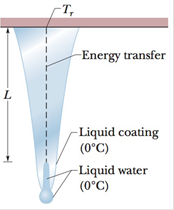
Figure 18-55 Problem 78.
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Additional Science Textbook Solutions
Modern Physics
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Physics: Principles with Applications
Physics for Scientists and Engineers, Technology Update (No access codes included)
Conceptual Physical Science (6th Edition)
Physics for Scientists and Engineers
- An aluminum rod 0.500 m in length and with a cross sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 3(H) K. (a) If one-halt of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool, (b) If the circular surface of the upper end of the rod is maintained at 300 K. what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 YV/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forwardA scuba diver has his lungs filled to half capacity (3 liters) when 10 m below the surface. If the diver holds his breath while quietly rising to the surface, what will the volume of the lungs be (in liters) at the surface? Assume the temperature is the same at all depths. (The density of water is 1.0x103 kg/m3arrow_forwardo42 0 A 20.0 g copper ring at 0.000°C has an inner diameter of D = 2.54000 cm. An aluminum sphere at 100.0°C has a diameter of d = 2.545 08 cm. The sphere is put on top of the ring (Fig. 18-36), and the two are allowed to come to thermal equilibrium, with no heat lost to the surroundings. The sphere just passes through the ring at the equilibrium tempera- ture. What is the mass of the Al Cu Figure 18-36 Problem 42. sphere?arrow_forward
- The average coefficient of linear expansion of copper is 17 x 10-6 (°C)-1 . The Statue of Liberty is 93 in tall on a summer morning when the temperature is 25°C. Assume the copper plates covering the statue are mounted edge to edge without expansion joints and do not buckle or bind on the framework supporting them as the day grows hot. What is the order of magnitude of the statue's increase in height? (a) 0.1 mm (b) 1 mm (c) 1 cm (d) 10 cm (e) 1 mlarrow_forwardWhen the hoof of a galloping horse hits the ground, the digital flexor tendon in its lower leg may stretch by 5% in length, a significant stretch for this 45 cm tendon. The tendon is elastic; most—but not all—of the energy stored in the stretch is returned, which allows for efficient locomotion. In an exercisinghorse, the tendon may reach a temperature of 46°C, much higher than the horse’s 38°C body temperature. How can you explain this temperature rise?arrow_forward..60 GO Figure Figure 18-46 shows the cross section of a wall made of three layers. The layer thicknesses are L₁, L2=0.700L₁, and L3=0.350L₁. The thermal conductivities are k₁, k₂ = 0.900k₁, and k3= 0.800k₁. The temperatures at the left side and right side of the wall are TH = 30.0°C and Tc = -15.0°C, respectively. Ther- mal conduction is steady. (a) What is the temperature difference AT₂ across layer 2 (between the left and right sides of the layer)? If k₂ were, instead, equal to 1.1k₁, (b) would the rate at which energy is con- ducted through the wall be greater than, less than, or the same as pre- viously, and (c) what would be the value of AT₂? TH k₁ k₂ kg Tc L₁ L2 L3 Figure 18-46 Problem 60. Air Ice Waterarrow_forward
- Two metal rods, one aluminum and one brass, are clamped down on one end, opposite to each other. At 0.0ºC, the rods are each 50.0 cm long and separated by 0.024 cm at their unfastened ends. At what temperature will the rods just come into contact? (Assume the base to which the rods are clamped undergoes a negligibly small thermal expansion.)arrow_forwardA pronghorn antelope can run at a remarkable 18 m/sm/s for up to 10 minutes, almost triple the speed that an elite human runner can maintain. For a 32 kgkg pronghorn, this requires an astonishing 3.4 kWkW of metabolic power, which leads to a significant increase in body temperature. If the pronghorn had no way to exhaust heat to the environment, by how much would its body temperature increase during this run? (In fact, it will lose some heat, so the rise won't be this dramatic, but it will be quite noticeable, requiring adaptations that keep the pronghorn's brain cooler than its body in such circumstances.) Assume the efficiency of the pronghorn to be equal to that of human.arrow_forward11) A brass rod is 69.5 cm long and an aluminum rod is 49.3 cm long when both rods are at an initial temperature of 0° C. The rods are placed in line with a gap of 1.2 cm between them, as shown in the figure. The distance between the far ends of the rods is maintained at 120.0 cm throughout. The temperature of both rods is raised equally until they are barely in contact. At what temperature does contact occur? The coefficients of linear expansion of brass and aluminum are 2.0 x10-5 K-1 (brass) and 2.4 x 10-5 K-1 (aluminum). -120.0 cm C) 510°C D) 470°C E) 440°C brass A) 490°C J!!!!! aluminum B) 420°Carrow_forward
- Ice of mass 10.0 kg at 0.00° C is placed in an ice chest. The ice chest has 2.00 cm thick walls of thermal coductivity 1.00 x 10^-5 kcal/s-m-C° and a surface area of 1.30m^2. (a) How much heat must be absorbed by the ice before it melts? (b) If the outer surface of the ice chest is at 30.0° C, how long will it take for the ice to melt?arrow_forward38 A 0.530 kg sample of liquid water and a sample of ice are placed in a thermally insulated container. The container also contains a device that transfers energy as heat from the liquid water to the ice at a constant rate P, until thermal equilibriumarrow_forwardA steel rod is initially 1 meter long at a temperature of 20°C. Assuming that the expansion is uniform along the entire length of the rod , if the coefficient of linear expansion for steel is 11 x 10^-6 per degree Celsius, calculate: a) The change in length of the rod when its temperature is increased to 100°C. b) The final length of the rod at 100°C.arrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
