
Fundamentals of Physics, Volume 1, Chapter 1-20
10th Edition
ISBN: 9781118233764
Author: David Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 8Q
For which cycle in Fig. 18-27, traversed clockwise, is (a) W greater and (b) Q greater?
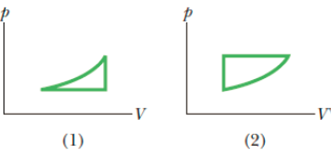
Figure 18-27 Questions 7 and 8.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(I) Solar cells (Fig. 15–26) can produce about 40 W of elec-
tricity per square meter of surface area if directly facing the
Sun. How large an area is required to supply the needs of
a house that requires 24 kWh/day? Would this fit on the
roof of an average house? (Assume the Sun shines about
9h/day.)
FIGURE 15-26
Problem 53.
..47 SSM www When a system is taken from state i to state f
along path iaf in Fig. 18-40, Q = 50 cal and W = 20 cal. Along path
ibf, Q = 36 cal. (a) What is W along path ibf? (b) If W = -13 cal
for the return path fi, what is Q for this path? (c) If Eint,i = 10 cal,
what is Eint,f? If Eint,b = 22 cal, what is Q for (d) path ib and (e)
path bf?
Pressure
a
Volume
Figure 18-40 Problem 47.
b
..47 SSM www When a system is taken from state i to state f
along path iaf in Fig. 18-40, Q = 50 cal and W = 20 cal. Along path
ibf, Q = 36 cal. (a) What is W along path ibf? (b) If W = -13 cal
for the return path fi, what is Q for this path? (c) If Eint,i = 10 cal,
what is Eint,f? If Eint,b = 22 cal, what is Q for (d) path ib and
(e) path bf?
Pressure
0
a
b
Volume
Figure 18-40 Problem 47.
Chapter 18 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Ch. 18 - The initial length L, change in temperature T, and...Ch. 18 - Figure 18-24 shows three linear temperature...Ch. 18 - Materials A, B, and C are solids that are at their...Ch. 18 - A sample A of liquid water and a sample B of ice,...Ch. 18 - Question 4 continued: Graphs b through f of Fig....Ch. 18 - Figure 18-26 shows three different arrangements of...Ch. 18 - Figure 18-27 shows two closed cycles on p-V...Ch. 18 - For which cycle in Fig. 18-27, traversed...Ch. 18 - Three different materials of identical mass are...Ch. 18 - A solid cube of edge length r, a solid sphere of...
Ch. 18 - A hot object is dropped into a thermally insulated...Ch. 18 - Suppose the temperature of a gas is 373.15 K when...Ch. 18 - Two constant-volume gas thermometers are...Ch. 18 - A gas thermometer is constructed of two...Ch. 18 - a In 1964, the temperature in the Siberian village...Ch. 18 - At what temperature is the Fahrenheit scale...Ch. 18 - On a linear X temperature scale, water freezes at...Ch. 18 - ILW Suppose that on a linear temperature scale X,...Ch. 18 - At 20C, a brass cube has edge length 30 cm. What...Ch. 18 - ILW A circular hole in an aluminum plate is 2.725...Ch. 18 - An aluminum flagpole is 33 m high. By how much...Ch. 18 - Prob. 11PCh. 18 - An aluminum-alloy rod has a length of 10.000 cm at...Ch. 18 - SSM Find the change in volume of an aluminum...Ch. 18 - When the temperature of a copper coin is raised by...Ch. 18 - ILW A steel rod is 3.000 cm in diameter at 25.00C....Ch. 18 - When the temperature of a metal cylinder is raised...Ch. 18 - SSM WWW An aluminum cup of 100 cm3 capacity is...Ch. 18 - At 20C, a rod is exactly 20.05 cm long on a steel...Ch. 18 - GO A vertical glass tube of length L = 1.280 000 m...Ch. 18 - GO In a certain experiment, a small radioactive...Ch. 18 - SSM ILW As a result of a temperature rise of 32 C,...Ch. 18 - One way to keep the contents of a garage from...Ch. 18 - SSM A small electric immersion healer is used to...Ch. 18 - A certain substance has a mass per mole of 50.0...Ch. 18 - Prob. 25PCh. 18 - What muss of butter, which has a usable energy...Ch. 18 - SSM Calculate the minimum amount of energy, in...Ch. 18 - How much water remains unfrozen after 50.2 kJ is...Ch. 18 - In a solar water heater, energy from the Sun is...Ch. 18 - A 0.400 kg simple is placed in a cooling apparatus...Ch. 18 - ILW What mass of steam at 100C must be mixed with...Ch. 18 - The specific heat of a substance varies with...Ch. 18 - Nonmetric version: a How long does a 2.0 105...Ch. 18 - GO Samples A and B are at different initial...Ch. 18 - An insulated Thermos contains l30 cm3 of hot...Ch. 18 - A 150 g copper bowl contains 220 g of water, both...Ch. 18 - A person makes a quantity of iced tea by mixing...Ch. 18 - A 0.530 kg sample of liquid water and a sample of...Ch. 18 - GO Ethyl alcohol has a boiling point of 78.0C, a...Ch. 18 - GO Calculate the specific heat of a metal from the...Ch. 18 - SSM WWW a Two 50 g ice cubes are dropped into 200...Ch. 18 - GO A 20.0 g copper ring at 0.000C has an inner...Ch. 18 - In Fig. 18-37, a gas sample expands from V0 to...Ch. 18 - GO A thermodynamic system is taken from stale A to...Ch. 18 - SSM ILW A gas within a closed chamber undergoes...Ch. 18 - Suppose 200 J of work is done on a system and 70.0...Ch. 18 - Prob. 47PCh. 18 - GO As a gas is held within a closed chamber, it...Ch. 18 - GO Figure 18-42 represents a closed cycle for a...Ch. 18 - GO A lab sample of gas is taken through cycle abca...Ch. 18 - A sphere of radius 0.500 m, temperature 27.0C, and...Ch. 18 - The ceiling of a single-family dwelling in a cold...Ch. 18 - SSM Consider the slab shown in Fig. 18-18. Suppose...Ch. 18 - If you were to walk briefly in space without a...Ch. 18 - ILW A cylindrical copper rod of length 1.2 m and...Ch. 18 - The giant hornet Vespa mandarinia japonica preys...Ch. 18 - Prob. 57PCh. 18 - A solid cylinder of radius r1 = 2.5 cm, length h1...Ch. 18 - Prob. 59PCh. 18 - GO Figure 18-46 shows the cross section of a wall...Ch. 18 - SSM A 5.0 cm slap has formed on an outdoor tank of...Ch. 18 - Leidenfrost effect. A water drop will last about 1...Ch. 18 - GO Figure 18-49 shows in cross section a wall...Ch. 18 - Prob. 64PCh. 18 - Ice has formed on a shallow pond, and a shady...Ch. 18 - GO Evaporative cooling of beverages. A cold...Ch. 18 - In the extrusion of cold chocolate from a tube,...Ch. 18 - Prob. 68PCh. 18 - Figure 18-51 displays a closed cycle for a gas....Ch. 18 - In a certain solar house, energy from the Sun is...Ch. 18 - A 0.300 kg sample is placed in a cooling apparatus...Ch. 18 - The average rate at which energy is conducted...Ch. 18 - What is the volume increase of an aluminum cube...Ch. 18 - In a series of experiment, block B is to be placed...Ch. 18 - Figure 18-54 displays a dosed cycle for a gas....Ch. 18 - Three equal-length straight rods, of aluminum,...Ch. 18 - SSM The temperature of a 0.700 kg cube of ice is...Ch. 18 - GO Icicles. Liquid water coats an active growing...Ch. 18 - SSM A sample of gas expands from an initial...Ch. 18 - Figure 18-56a shows a cylinder containing gas and...Ch. 18 - SSM A sample of gas undergoes a transition from an...Ch. 18 - Prob. 82PCh. 18 - SSM The temperature of a Pyrex disk is changed...Ch. 18 - a Calculate the rate at which body heat is...Ch. 18 - SSM A 2.50 kg Jump of aluminum is heated to 92.0C...Ch. 18 - A glass window pane is exactly 20 cm by 30 cm at...Ch. 18 - A recruit can join the semi-secret 300 F club at...Ch. 18 - A steel rod at 25.0C is bolted at both ends and...Ch. 18 - An athlete needs to lose weight and decides to do...Ch. 18 - Soon after Earth was formed, heat released by the...Ch. 18 - Prob. 91PCh. 18 - A rectangular plate of glass initially has the...Ch. 18 - Suppose that you intercept 5.0 103 of the energy...Ch. 18 - A thermometer of mass 0.0550 kg and of specific...Ch. 18 - A sample of gas expands from V1 = 1.0 m3 and p1 =...Ch. 18 - Figure 18-59 shows a composite bar of length L =...Ch. 18 - On finding your stove out of order, you decide to...Ch. 18 - The p-V diagram in the Fig. 18-60 shows two paths...Ch. 18 - A cube of edge length 6.0 106 m, emissivity 0.75,...Ch. 18 - A flow calorimeter is a device used to measure the...Ch. 18 - An object of mass 6.00 kg falls through a height...Ch. 18 - The Pyrex glass mirror in a telescope has a...Ch. 18 - The area A of a rectangular plate is ab = 1.4 m2....Ch. 18 - Consider the liquid in a barometer whose...Ch. 18 - A pendulum clock with a pendulum made of brass is...Ch. 18 - Prob. 106PCh. 18 - Prob. 107PCh. 18 - A 1700 kg Buick moving at 83 km/h brakes to a...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Find the rate of conduction heat transfer per unit area through a 2 in. thick brick, k=0.4Btu/h-ft-R , with a t...
EBK FUNDAMENTALS OF THERMODYNAMICS, ENH
(a) Find the work done in lifting 1 L of blood (mass 1 kg) from the foot to the head of a 1.7-m-tall person. (b...
Essential University Physics: Volume 1 (3rd Edition)
The magnitude of the electric field.
Sears And Zemansky's University Physics With Modern Physics
Q8.13 In Fig. 8.23b, the kinetic energy of the Ping-Pong ball is larger after its interaction with the bowling ...
University Physics with Modern Physics (14th Edition)
Consider hydrocarbon molecules with the formula C4Hx in which the four carbons form a linear chain. Sketch the ...
MODERN PHYSICS (LOOSELEAF)
13. What happens to a nitrogen atom in the atmosphere that captures a neutron?
Conceptual Physical Science (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Compare the charge in internal energy of an ideal gas for a quasi-static adiabatic expansion with that for a quasi-static isothermal expansion. What happens to the temperature of an ideal gas in an adiabatic expansion?arrow_forwardShow that the coefficients of performance of refrigerators and heat pumps are related by COPref=COPhp1. Start with the definitions of the COP s and the conservation of energy relationship between Qh, QC, and W.arrow_forwardIn a diesel engine, the fuel is ignited without a spark plug. Instead, air in a cylinder is compressed adiabatically to a temperature above the ignition temperature of the fuel; at the point of maximum compression, the fuel is injected into the cylinder. Suppose that air at 20 C is taken into the cylinder at a volume V1 and then compressed adiabatically and quasi-statically to a temperature of 600 C and a volume V2 . If =1.4 , what is the ratio V1/V2 ? (Note: static. In an operating diesel engine, the compression is not quasi-arrow_forward
- Module 18-5 The First Law of Thermodynamics •43 In Fig. 18-37, a gas sample ex- pands from Vo to 4.0Vo while its pressure decreases from Po to Po/4.0. If Vo =1.0 m³ and Po = 40 Pa, how much work is done by the gas if its pressure changes with vol- ume via (a) path A, (b) path B, and (c) path C? %3! V% 4.0V, Pressure (Pa)arrow_forward47 SSM www When a system is taken from state i to state f along path iaf in Fig. 18-40, Q = 50 cal and W = 20 cal. Along path ibf, Q = 36 cal. (a) What is W along path ibf? (b) If W = -13 cal for the return path fi, what is Q for this path? (c) If Einti = 10 cal, what is Ent ? If Ent = 22 cal, what is Q for (d) path ib and (e) path bf? Volume Figure 18-40 Problem 47. Pressurearrow_forward87 Figure 19-29 shows a cycle consisting of five paths: AB is isothermal at 300 K, BC is adiabatic with work = 5.0 J, CD is at a constant pressure of 5 atm, DE is isothermal, and EA is adiabatic with a change in internal energy of 8.0 J. What is the change in in- ternal energy of the gas along path CD?arrow_forward
- Calculate q when 29 J of work is done on a system and its internal energy increases by 96 J.arrow_forwardIt is important for the body to be able to cool itself even during moderate exercise. Suppose a 70kg man runs on a treadmill for 30min and at a (total) metabolic energy expenditure rate of 1000W. Remembering that the human body has a 25% efficiency, estimate the amount of (mechanical) work done on the treadmill during that time. A) 450kJB)1350kJC)1000kJD)1800kJarrow_forward48 Go As a gas is held within a closed chamber, it passes through the cycle shown in Fig. 18-41. Determine the energy transferred by the system as heat during constant-pressure process CA if the energy added as heat QAB during constant-volume process AB is 20.0 J, no energy is transferred as heat during adiabatic process BC, and the net work done during the cycle 'C Volume is 15.0 J. Figure 18-41 Problem 48. Pressurearrow_forward
- (17 blanks) Calculate the efficiency of the Carnot Engine for ideal gas. Consider the figure below for a complete cycle of Carnot engine, where the cycle goes from ad->ab->bc->cd Carnot cycle Он TH Ted 2016 Fearson Educaton, Inc. processes; and the volumes involved are Va, Vb, Vc. Vd, and the two processes and segment ab & cd are the Where segments ad & bc are the temperatures TC and Sol. Since AU = 0, then the heat on segment ab is QH = TH In |arrow_forward..48 GO As a gas is held within a closed chamber, it passes through the cycle shown in Fig. 18-41. Determine the energy transferred by the system as heat during constant-pressure process CA if the energy added as heat QAB during constant-volume process AB is 20.0 J, no energy is transferred as heat during adiabatic process BC, and the net work done during the cycle is 15.0 J. Pressure B A C 0 Volume Figure 18-41 Problem 48.arrow_forward17 In Fig. 20-25, where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscil- lating. What are (a) pz/p1- (b) P3/P1, and (c) T3/T? For path 1 2, what are (d) W/NRT,, (e) QinRT, (f) AEm/nRT, and (g) AS/nR? For path 2→ 3, what are (h) W/nRT, (i) QinRT,, (j) AEin/NRT1, (k) AS/nR? For path 3 → 1, what are (1) W/NRT,, (m) QinRT,, (n) AEin/nRT1, and (o) AS/nR? Isothermal Adiabatic 2. Vas Vị Volume Figure 20-25 Problem 17. Pressurearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College


College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY