
Concept explainers
Rank the compounds in each group in order of increasing basicity.
a.
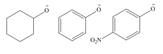
b.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry-Package(Custom)
- Question 5 5. Rank each of the five compounds by relative basicity. Use a "1" for the strongest base, a "2" for the 2nd strongest, all the way to a "5" for the weakest base. NH2 ZHN NH2 Meo NH2 NH2 O2Narrow_forward2. Which of the following is most basic? Which is least basic? NH₂ WHECH 3arrow_forwardArrange the given compounds based on their relative Brønsted acidities. H-F H-Br H-SH H-CH3 H-NH2arrow_forward
- Rank the given compounds in order of decreasing basicity. 1=most basic, 4=least basicarrow_forwardRank the following species in order of increasing basicity. Explain your reasons for ordering them as you do.NH3 CH3O- H2O CH3COO- NaOH NH - 2 HSO4arrow_forwardA ΝΗ most basic B > V с NH₂ V NH₂ H3CO E least basic NH₂arrow_forward
- 5. Rank each of the five compounds by relative basicity. Use a “1" for the strongest base, a "2" for the 2nd strongest, all the way to a "5" for the weakest base. NH2 .NH2 NH2 Мео NH2 NH2 O2Narrow_forwardWhich is a stronger base?a. HS− or HO− c. CH3OH or CH3O−d. Cl− or Br− b. CH3O− or CH3NH − CH3COO− or CF3COO−CH3CHClCOO− or CH3CHBrCOO−arrow_forwardRank the compounds in each group in order of increasing acidity. ÇI Br .COOH „COOH COOH а. COOH COOH COOH CH CF3 LOH HO HO HO HO HO b. d. CH O,N Br O,N O,N `NO2 Rank the compounds in each group in order of increasing basicity. NH2 NH2 NH2 a. NH3 -NH2 -NH2 c. O,N CH3 b. d. CgHgNH2 (C,H5)2NH -NH2 N. H H C.arrow_forward
- Rank the following in order of increasing basicity. The first in your list should be the least basic. HO 1 0 3 2 < 1 O2 3<1 01 2<3 01 3<2 3 1< 2 3. 2.arrow_forwardRank these compounds from weakest to strongest acid H3C I ОН F3C. OI II ОН F3C III ОН Cl3 C IV ОНarrow_forwardWrite "highest" under the compound with the highest boiling point. Write "lowest" under the compound with the lowest boiling point. 1,2,3-propanetriol 1-pentanol 1,4-butanediol Write "most" under the compound which is most acidic. Write "least" under the compound which is least acidic. OH OH OH H.C HyCarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

