
Organic Chemistry-Package(Custom)
4th Edition
ISBN: 9781259141089
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.14P
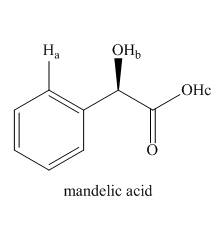
Rank the labeled protons
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Glycolic acid, HOCH2CO2H, is the simplest member of a group of compounds called αhydroxy acids, ingredients in skin care products that have an OH group on the carbon adjacent to a CO2H group. Would you expect HOCH2CO2H to be a stronger or weaker acid than acetic acid, CH3CO2H?
2-Hydroxybutanedioic acid occurs naturally in apples and other fruits. Rank the labeled protons in 2-hydroxybutanedioic acid in order of increasing acidity.
3, Which of the molecules drawn below is most acidic? Draw the conjugate base and explain
thoroughly.
वे देई
d
a
C
b
Chapter 19 Solutions
Organic Chemistry-Package(Custom)
Ch. 19 - Give the IUPAC name for each compound.Ch. 19 - Problem 19.2 Give the structure corresponding to...Ch. 19 - Problem 19.3 Draw the structure corresponding to...Ch. 19 - Prob. 19.4PCh. 19 - Prob. 19.5PCh. 19 - Rank the following compounds in order of...Ch. 19 - Explain how you could use IR spectroscopy to...Ch. 19 - Identify the structure of a compound of molecular...Ch. 19 - Problem 19.9 How many tetrahedral stereogenic...Ch. 19 - What alcohol can be oxidized to each carboxylic...
Ch. 19 - Prob. 19.11PCh. 19 - Draw the products of each acid-base reaction.Ch. 19 - Problem 19.14 Given the values in Appendix A,...Ch. 19 - Problem 19.15 Rank the labeled protons in...Ch. 19 - Problem 19.16 Match each of the following values ...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - Prob. 19.18PCh. 19 - Which of the following pairs of compounds can be...Ch. 19 - Problem 19.21 Two other commonly used sulfonic...Ch. 19 - Problem 19.22 Draw both enantiomers of each amino...Ch. 19 - Problem 19.23 Explain why amino acids, unlike most...Ch. 19 - Problem 19.24 Draw the positively charged,...Ch. 19 - Prob. 19.24PCh. 19 - Problem 19.26 Explain why the of the group of...Ch. 19 - Answer each question for A and B depicted in the...Ch. 19 - Prob. 19.27PCh. 19 - Give the IUPAC name for each compound. a....Ch. 19 - Prob. 19.29PCh. 19 - Draw the structures and give the IUPAC names for...Ch. 19 - Prob. 19.31PCh. 19 - Rank the compounds in each group in order of...Ch. 19 - 19.33 Draw the organic products formed in each...Ch. 19 - 19.34 Identify the lettered compounds in each...Ch. 19 - 19.35 Using the table in Appendix A, determine...Ch. 19 - Draw the products of each acid-base reaction, and...Ch. 19 - Which compound in each pair has the lower pKa?...Ch. 19 - 19.38 Rank the compounds in each group in order of...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - 19.40 Match the values to the appropriate...Ch. 19 - Prob. 19.41PCh. 19 - 19.42 Which carboxylic acid has the lower ,...Ch. 19 - Prob. 19.43PCh. 19 - 19.44 Explain the following statement. Although...Ch. 19 - Prob. 19.45PCh. 19 - 19.46 Explain why the of compound A is lower than...Ch. 19 - 19.47 Rank the following compounds in order of...Ch. 19 - Explain the following result. Acetic acid...Ch. 19 - 19.50 Draw all resonance structures of the...Ch. 19 - As we will see in Chapter 23, CH bonds are...Ch. 19 - Prob. 19.51PCh. 19 - The pKa of acetamide (CH3CONH2) is 16. Draw the...Ch. 19 - 19.54 Write out the steps needed to separate...Ch. 19 - Prob. 19.54PCh. 19 - Can octane and octan -1- ol be separated using an...Ch. 19 - 19.57 Identify each compound from its spectral...Ch. 19 - 19.58 Use the NMR and IR spectra given below to...Ch. 19 - 19.59 An unknown compound (molecular formula )...Ch. 19 - 19.60 Propose a structure for (molecular formula...Ch. 19 - Prob. 19.60PCh. 19 - 19.61 Match the data to the appropriate...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.64PCh. 19 - 19.65 For each amino acid ,draw its neutral,...Ch. 19 - Calculate the isoelectric point for each amino...Ch. 19 - 19.67 Lysine and tryptophan are two amino acids...Ch. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - Prob. 19.70PCh. 19 - Prob. 19.71PCh. 19 - 19.71 Hydroxy butanedioic acid occurs naturally in...Ch. 19 - 19.72 Although it was initially sold as a rat...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Answer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forwardWhich has the larger numerical value? (a) The pKa of a strong acid or the pKa of a weak acid (b) The Ka of a strong acid or the Ka of a weak acidarrow_forwardA very strong base can remove a proton from methylamine:arrow_forward
- Arrange the following compounds in order of decreasing acidity. I || > ||| > | O III >> || ||| > || > | O I > I| > ||| CH₂ II III 0arrow_forwardDraw the structure for the conjugate acid of the base shown. Be sure to include all lone pairs and formal charges (if applicable) on the structure. H Harrow_forwardWhich is the stronger base, CH3PH or CH3S? Hint: Which base has the weaker conjugate acid? CH3-S: VS IA Η | ΠΙΑ LBe 11 12 Na Mg CH3-PH IIIA IVA VA VIA VILA He 9 10 B 68 80 104 106 106 107 108 100 110 Fr Ra Ac Ung Unp Unh Uns Uno Une Unn Select one: O A. CH3PH* O B. CH3S™ 51 13 Al 14 CNO F Ne Si 15 16 S "a P 37 38 30 40 41 42 43 44 45 46 47 48 40 50 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te VIILA 66 57 56 72 73 74 75 76 77 78 791 80 81 82 83 Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi 22 23 24 25 26 27 28 29 30 31 32 -33 34 35 19 20 21 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 20 By By 30 E 17 CI Ar 18 61 62 63 54 I Xe 84 85 Po At Rn 86arrow_forward
- Which base, CH3NH2 or (CH3)2NH, is the stronger base? Which conjugate acid, (CH3)2 NH2+ or(CH3)2 NH3+ , is the stronger acid?arrow_forwardIn each row of the table, select the stronger base. HO NH to alo CH3 N_ CI N_ OHarrow_forwardChoose the strongest base. Digion OH3C-NH₂ NH₂ NH₂ NO₂ NH₂arrow_forward
- Consider the two following nitrophenol structures: OH OH `NO2 NO2 p-nitrophenol m-nitrophenol Predict which will be the stronger acid. (Hint: Consider possible resonance structures analogous to those given in the text for phenol.)arrow_forwardH2SO4 + NH3 ==== HSO4- + NH4+ . Which of the products is a conjugate base?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning



General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY