
Concept explainers
(a)
Interpretation:
The
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is
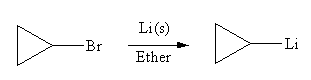
Explanation of Solution
The given reaction is,
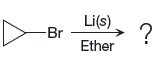
In the above reaction, the C atom of the C-Br bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. An alkyllithium reagent (RLi) can be synthesized from an alkyl bromide by treating it with solid lithium in ether. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:
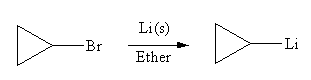
The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(b)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. An alkyl bromide can be converted to a Grignard reagent (RMgBr) simply by treating it with solid magnesium in an ether solvent such as tetrahydrofuran (THF).
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
Explanation of Solution
The given reaction is
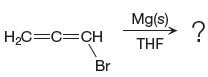
In the above reaction, the C atom of the C-Br bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. An alkyl bromide can be converted to a Grignard reagent (RMgBr) simply by treating it with solid magnesium in an ether solvent such as tetrahydrofuran (THF). So the C-Br bond will become C-Mg bond. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:

The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(c)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. An alkyl bromide can be converted to a Grignard reagent (RMgBr) simply by treating it with solid magnesium in an ether solvent such as tetrahydrofuran (THF).
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
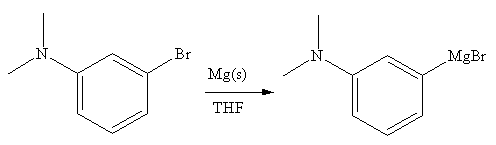
Explanation of Solution
The given reaction is
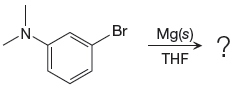
In the above reaction, the C atom of the C-Br bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. An alkyl bromide can be converted to a Grignard reagent (RMgBr) simply by treating it with solid magnesium in an ether solvent such as tetrahydrofuran (THF). So the C-Br bond will become C-Mg bond. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:

The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(d)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. An alkyl bromide can be converted to a Grignard reagent (RMgX) simply by treating it with solid magnesium in an ether solvent.
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
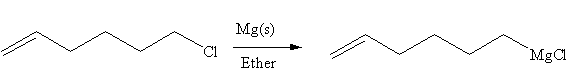
Explanation of Solution
The given reaction is
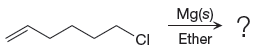
In the above reaction, the C atom of the C-Cl bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. An alkyl chloride can be converted to a Grignard reagent (RMgBr) simply by treating it with solid magnesium in an ether. So the C-Cl bond will become C-Mg bond. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:
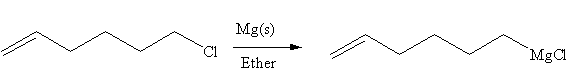
The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(e)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. A lithium dialkylcuprate is synthesized from the corresponding alkyllithium reagent by treating it with copper(I) iodide, CuI.
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
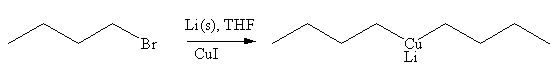
Explanation of Solution
The given reaction is
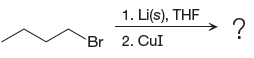
In the above reaction, the C atom of the C-Br bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. Lithium dialkylcuprate is synthesized from the corresponding alkyllithium reagent by treating it with copper(I) iodide, CuI. So the C-Br bond will become C-CuLi bond. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:

The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(f)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. An alkyllithium reagent (RLi) can be synthesized from an alkyl bromide by treating it with solid lithium in ether solvent such as THF.
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
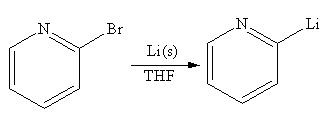
Explanation of Solution
The given reaction is
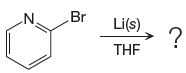
In the above reaction, the C atom of the C-Br bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. An alkyllithium reagent (RLi) can be synthesized from an alkyl bromide by treating it with solid lithium in ether solvent such as THF. So the C-Br bond will become C-Li bond. Therefore the organometallic compound that would be produced by the given reaction is as shown below:

The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
(g)
Interpretation:
The organometallic compound that would be produced by the given reaction is to be drawn.
Concept introduction:
To form a bond between two carbons of like charge, a separate reaction that first reverses the charge (or polarity) at one of the carbons is carried out. Thus, one carbon atom would become electron rich while the other would remain electron poor. This general idea of reversing a charge at a particular atom is called umpolung. In the alkyl halide reactant, the C atom bonded to the halogen atom bears a partial positive charge and is relatively electron poor. By contrast, that C atom has to become electron rich in the organometallic compound produced. A lithium dialkylcuprate is synthesized from the corresponding alkyllithium reagent by treating it with copper(I) iodide, CuI.
Answer to Problem 19.41P
The organometallic compound that would be produced by the given reaction is shown below:
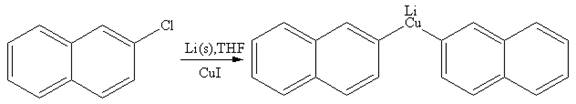
Explanation of Solution
The given reaction is
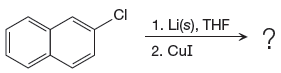
In the above reaction, the C atom of the C-Cl bond is electron-poor. When the C-Br bond is bonded to the metal, the carbon will become electron-rich. A lithium dialkylcuprate is synthesized from the corresponding alkyllithium reagent by treating it with copper(I) iodide, CuI. Therefore, the organometallic compound that would be produced by the given reaction is as shown below:

The organometallic compound that would be produced by the given reaction is drawn by converting the electron-poor carbon of C-Br bond to an electron-rich carbon.
Want to see more full solutions like this?
Chapter 19 Solutions
Get Ready for Organic Chemistry
- Draw the mechanism arrows for the reaction between an acid chloride and an alcohol.arrow_forwardWhich of the following is the rule which states that the more substituted product is the major product in an elimination reaction? O 1) Boyle's Law O 2) Markovnikov's Rule 3) Zaitsev's Rule O 4) LeChatlier's Principlearrow_forwardThe following reaction can be classified as: elimination acid rearrangement substitution additionarrow_forward
- What is the effect on the rate constant and reaction rate when reaction temperature is increased? The rate constant will be greater, and the reaction rate will be higher. The rate constant will be smaller, but the reaction rate will be higher. The rate constant will be smaller, and the reaction rate will be lower.arrow_forwardWhen this compound is treated with a base, elimination occurs. What is the product of this elimination? Explain your reasoning.arrow_forwardPlease draw a detailed mechanism for all steps in the following reaction sequence. Please clearly show electron movement.arrow_forward
- In an electrophilic aromatic substitution, the substitution pattern on the product is determine by the directing nature of the substituents on the ring. What controls the substitution pattern in a nucleophilic aromatic substitution reaction?arrow_forwardComplete the following reaction and provide the detailed mechanism NaOH NaOH +arrow_forwardThe compound shown here is the product of a base-catalyzed aldol condensation. Draw the structure of the nonaromatic-containing starting material that is present before the aldol condensation and the aldol addition.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



