
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.76P
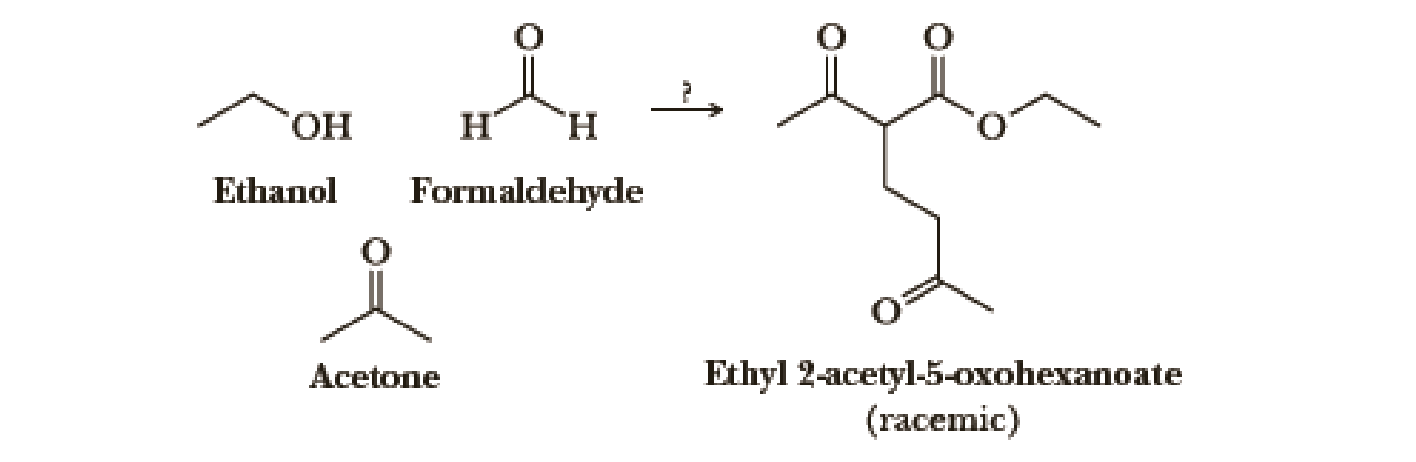
Using your reaction roadmaps as a guide, show how to convert ethanol, formaldehyde, and acetone into racemic ethyl 2-acctyl-5-oxohcxanoatc. You must use ethanol, formaldehyde, and acetone as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Show how to convert (E)-hex-3-ene into propyl propionate. You must use (E)-hex-3-ene as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.
A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueoussolution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.
Provide the reagents necessary to convert (R)-2-chlorobutane to (S)-2-methoxybutane.
Chapter 19 Solutions
Organic Chemistry
Ch. 19.2 - Prob. 19.1PCh. 19.2 - Prob. 19.2PCh. 19.2 - Prob. 19.3PCh. 19.3 - Prob. 19.4PCh. 19.3 - Prob. 19.5PCh. 19.3 - Prob. 19.6PCh. 19.5 - Prob. 19.7PCh. 19.5 - Prob. 19.8PCh. 19.5 - Prob. 19.9PCh. 19.6 - Prob. 19.10P
Ch. 19.6 - Prob. 19.11PCh. 19.7 - Prob. 19.12PCh. 19.8 - Prob. 19.13PCh. 19.8 - Prob. 19.14PCh. 19.8 - Prob. 19.15PCh. 19.8 - Prob. 19.16PCh. 19.9 - Prob. 19.17PCh. 19.9 - Prob. AQCh. 19.9 - Prob. BQCh. 19.9 - Prob. CQCh. 19.9 - Prob. DQCh. 19.9 - Prob. EQCh. 19.9 - Prob. FQCh. 19.9 - Prob. GQCh. 19.9 - Intermediate G in Synthesis III is produced as a...Ch. 19.9 - Prob. IQCh. 19.9 - Prob. JQCh. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - Prob. 19.20PCh. 19 - Prob. 19.21PCh. 19 - Prob. 19.22PCh. 19 - Prob. 19.23PCh. 19 - Cyclohexene can be converted to...Ch. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - Prob. 19.28PCh. 19 - Prob. 19.29PCh. 19 - Prob. 19.30PCh. 19 - Draw structural formulas for the -ketoesters...Ch. 19 - Prob. 19.32PCh. 19 - Prob. 19.33PCh. 19 - Propose a synthesis for each ketone, using as one...Ch. 19 - Prob. 19.35PCh. 19 - Claisen condensation between diethyl phthalate and...Ch. 19 - Prob. 19.37PCh. 19 - Prob. 19.38PCh. 19 - Prob. 19.39PCh. 19 - Enamines normally react with methyl iodide to give...Ch. 19 - Prob. 19.41PCh. 19 - Prob. 19.42PCh. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49PCh. 19 - Prob. 19.50PCh. 19 - Prob. 19.51PCh. 19 - Prob. 19.52PCh. 19 - Show experimental conditions by which to carry out...Ch. 19 - Prob. 19.55PCh. 19 - The compound 3,5,5-trimethyl-2-cyclohexenone can...Ch. 19 - Prob. 19.57PCh. 19 - Prob. 19.58PCh. 19 - The widely used anticoagulant warfarin (see...Ch. 19 - Following is a retrosynthetic analysis for an...Ch. 19 - Following are the steps in one of the several...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.65PCh. 19 - Prob. 19.67PCh. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - In Problem 7.28, we saw this two-step sequence in...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Using your reaction roadmaps as a guide, show how...Ch. 19 - Prob. 19.79PCh. 19 - Prob. 19.80PCh. 19 - Prob. 19.81PCh. 19 - The following molecule undergoes an intramolecular...
Additional Science Textbook Solutions
Find more solutions based on key concepts
The smallest building blocks inside your cell phone are about 1000 times smaller than the diameter of a human h...
Chemistry In Context
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, & Biological Chemistry
Real walls are never totally adiabatic. Use your experience to order the following walls in increasing order wi...
Thermodynamics, Statistical Thermodynamics, & Kinetics
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. Show all required reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert (2-methylpropyl) benzene into 4-phenyl-3-buten-2-one. You must use (2-methylpropyl) benzene as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 3-hexyne into propanal. All of the carbon atoms of the target molecule must be derived from the starting material as efficiently as possible. Show all intermediate molecules synthesized along the way.arrow_forward
- Using your reaction roadmap as a guide, show how to convert 1-bromopentane and sodium cyanide into N-hexylhexanamide. You must use 1-bromopentane and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert ethanol into 2-pentanone. You must use ethanol as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-bromopropane and carbon dioxide into 4-heptanone. You must use 1-bromopropane and carbon dioxide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forward
- Using your reaction roadmaps as a guide, show how to convert 1-butanol into racemic 4-octanol. You must use 1-butanol as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert cyclohexane and ethanol into racemic ethyl 2-oxocyclopentanecarboxylate. You must use ethanol and cyclohexane as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your old and new reaction roadmaps as a guide, show how to convert 1-bromo-3-methylbutane into 2, 7-dimethyloctane. You must use l-bromo-3-methylbutane as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forward
- Following is a retrosynthetic analysis for the synthesis of the herbicide (S)-Metolachlor from 2-ethyl-6-methylaniline, chloroacetic acid, acetone, and methanol. Show reagents and experimental conditions for the synthesis of Metolachlor from these four organic starting materials. Your synthesis will most likely give a racemic mixture. The chiral catalyst used by Novartis for reduction in Step 2 gives 80% enantiomeric excess of the S enantiomer.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert ethylene into 2-methyl-1,3-dioxolane. You must use ethylene as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 2-methylpentane into 2-methyl-3-pentanone. Show all reagents needed and all molecules synthesized along the way.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY