
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
4th Edition
ISBN: 9780134465715
Author: John E. McMurry, David S. Ballantine
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.89GP
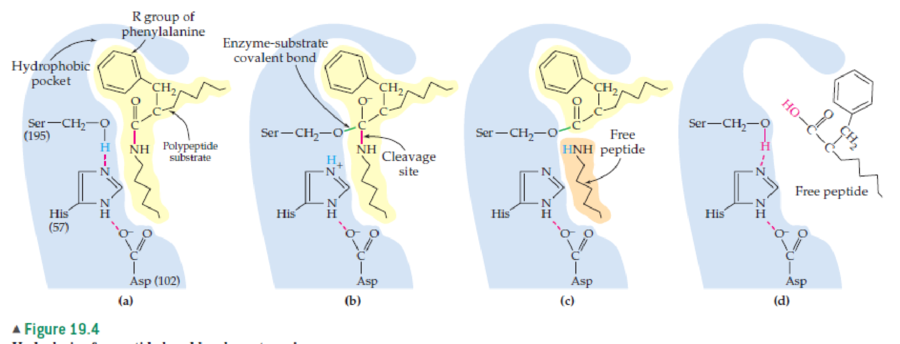
The ability to change a selected amino acid residue to another amino acid is referred to as “point mutation” by biochemists. Referring to the reaction for peptide bond hydrolysis in Figure 19.4, speculate on the effects that the following point mutations might have on the chymotrypsin mechanism shown in Figure 19.4: serine to valine; aspartate to glutamate.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Some of the following four amino acids :
alanine, arginine, histidine, aspartic acid would
provide a side chain for acid-base catalysis at
physiological pH (assume pK of each amino acid
is equal to pK value for the free amino acid in
solution).
Explain for each amino acid how and why each
would or would not provide the side chain
residue to support acid-base catalysis at
physiological pH.
Which of the following statements are descriptions of metal ion catalysis or
examples of metal ion catalysis? Choose all correct answers
a Zn²+ cofactor may properly orient the substrate in the active site through
ionic interactions.
a covalent bond forms between enzyme and substrate
lowers the energy or stabilizes the transition state or intermediate
catalyst retains its original form after reaction occurs
catalysts may participate in oxidation-reduction reactions by changes in the
oxidation state
H
CH₂
H₂C
HC-CH3
CH₂
H
H₂C
(S)
H₂C
H
CH₂
CH₂
CH₂
NH
O
C NH
NH₂
a) Which of the following statements about
this peptide are correct?
Group of answer choices
Treatment of this peptide with trypsin
generates two products.
This peptide is a substrate for
carboxypeptidase A
Treatment of this peptide with cyanogen
bromide generates a pentapeptide and a
tripeptide.
Treatment of this peptide with chymotrypsin
generates three products.
Treatment of this peptide with elastase
generates 2 products.
None of the above statements are correct.
b) What is the sequence of this peptide using
one letter abbreviations?
c) What is the pH which would correspond to
the ionization of the peptide as drawn above?
1, 5, 7, 10, 14
Chapter 19 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
Ch. 19.1 - Prob. 19.1PCh. 19.1 - The enzyme LDH converts lactate to pyruvate. In...Ch. 19.2 - The cofactors NAD+, Cu2+, Zn2+, coenzyme A, FAD,...Ch. 19.3 - Describe the reactions that you would expect these...Ch. 19.3 - Prob. 19.5PCh. 19.3 - Prob. 19.6PCh. 19.3 - Prob. 19.7PCh. 19.3 - Prob. 19.8PCh. 19.4 - Prob. 19.9KCPCh. 19.5 - Prob. 19.10KCP
Ch. 19.5 - Prob. 19.11PCh. 19.5 - Prob. 19.12PCh. 19.6 - Prob. 19.13PCh. 19.6 - Prob. 19.14PCh. 19.7 - (a) L-Threonine is converted to L-isoleucine in a...Ch. 19.8 - AZT (zidovudine) inhibits the synthesis of the HIV...Ch. 19.8 - Prob. 19.3CIAPCh. 19.8 - Prob. 19.16PCh. 19.9 - Does the enzyme described in each of the following...Ch. 19.9 - Prob. 19.18PCh. 19.9 - Compare the structures of vitamin A and vitamin C....Ch. 19.9 - Prob. 19.20PCh. 19.9 - Prob. 19.21KCPCh. 19.9 - Prob. 19.22PCh. 19.9 - Prob. 19.4CIAPCh. 19.9 - Prob. 19.6CIAPCh. 19.9 - Prob. 19.7CIAPCh. 19.9 - Enzyme levels in blood are often elevated in...Ch. 19.9 - Prob. 19.9CIAPCh. 19.9 - Prob. 19.23PCh. 19 - Prob. 19.24UKCCh. 19 - Prob. 19.25UKCCh. 19 - Prob. 19.26UKCCh. 19 - Prob. 19.27UKCCh. 19 - Prob. 19.28APCh. 19 - Explain how the following mechanisms regulate...Ch. 19 - Prob. 19.30APCh. 19 - Prob. 19.31APCh. 19 - Prob. 19.32APCh. 19 - Prob. 19.33APCh. 19 - Prob. 19.34APCh. 19 - Prob. 19.35APCh. 19 - Prob. 19.36APCh. 19 - Prob. 19.37APCh. 19 - Name an enzyme that acts on each molecule. (a)...Ch. 19 - Name an enzyme that acts on each molecule. (a)...Ch. 19 - What features of enzymes make them so specific in...Ch. 19 - Describe in general terms how enzymes act as...Ch. 19 - Prob. 19.42APCh. 19 - Prob. 19.43APCh. 19 - Prob. 19.44APCh. 19 - Prob. 19.45APCh. 19 - Prob. 19.46APCh. 19 - Prob. 19.47APCh. 19 - What is the difference between the lock-and-key...Ch. 19 - Why is the induced-fit model a more likely model...Ch. 19 - Prob. 19.50APCh. 19 - Prob. 19.51APCh. 19 - How do you explain the observation that pepsin, a...Ch. 19 - Prob. 19.53APCh. 19 - Prob. 19.54APCh. 19 - Prob. 19.55APCh. 19 - Prob. 19.56APCh. 19 - Prob. 19.57APCh. 19 - The text discusses three forms of enzyme...Ch. 19 - Prob. 19.59APCh. 19 - Prob. 19.60APCh. 19 - Prob. 19.62APCh. 19 - Prob. 19.63APCh. 19 - The meat tenderizer used in cooking is primarily...Ch. 19 - Prob. 19.65APCh. 19 - Why do allosteric enzymes have two types of...Ch. 19 - Prob. 19.67APCh. 19 - Prob. 19.68APCh. 19 - Prob. 19.69APCh. 19 - Prob. 19.70APCh. 19 - Prob. 19.71APCh. 19 - Prob. 19.72APCh. 19 - Prob. 19.73APCh. 19 - Prob. 19.74APCh. 19 - Prob. 19.75APCh. 19 - Prob. 19.76APCh. 19 - Prob. 19.77APCh. 19 - Prob. 19.78APCh. 19 - Prob. 19.79APCh. 19 - Prob. 19.80CPCh. 19 - Prob. 19.81CPCh. 19 - Prob. 19.82CPCh. 19 - Prob. 19.83CPCh. 19 - Prob. 19.84CPCh. 19 - Prob. 19.85CPCh. 19 - Prob. 19.86CPCh. 19 - Prob. 19.87CPCh. 19 - Prob. 19.88GPCh. 19 - The ability to change a selected amino acid...Ch. 19 - Prob. 19.90GPCh. 19 - Prob. 19.91GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- The endorphins are a group of naturally occurring neurotransmitters that act in a manner like morphine to control pain. Research has shown that the biologically active parts of the endorphin molecules are simple pentapeptides called enkephalins. Draw the structure of the methionine enkephalin with the sequence Tyr-Gly-Gly-Phe-Met. Identify the N-terminal and C-terminal amino acids. Bradykinin is a nonapeptide that stimulates smooth muscle contraction, dilates blood vessels, causes pain, and is a component of bee venom. Give the IUPAC name of the nonapeptide. Note: The selected photo is the sequence of Bradykinin.arrow_forwardthe following peptide is incubated as chymotrypsin: Gly-Val-Phe-Lys-Ala. Present the detailed chemical mechanism by which chymotrypsin hydrolyzes this peptide. Briefly explain each step. Include only the structure of the final products of the reaction.arrow_forwardAn enzyme that catalyzes disulfide– sulfhydryl exchange reactions, called protein disulfide isomerase (PDI), has been isolated. PDI rapidly converts inactive scrambled ribonuclease into enzymatically active ribonuclease. In contrast, insulin is rapidly inactivated by PDI. What does this important observation imply about the relation between the amino acid sequence of insulin and its threedimensional structure?arrow_forward
- Compare and contrast the biological roles of the following amino acids the following pairs ofamino acids. Once you have documented these role state which member of the pair is most important and why.arrow_forwardDraw the mechanism for the hydrolysis of the peptide bonds of aspartame. Draw all arrowsarrow_forwardSnake venom contains many hydrolase enzymes, including several serine proteases. One such protease (present in high concentrations in snake venom) mimics the action of thrombin to activate fibrin. The venom protease cleaves peptide bonds with a specificity for basic, positively charged amino acids. This enzyme is incubated with a substrate peptide with the sequence SNPDCKVALYMTW. Draw a mechanism for the peptide hydrolysis of this specific peptide catalyzed by this snake venom protease. Include the structure of the active site amino acids of the enzyme and the substrate peptide group (including the full structure of the 2 specific amino acids that contribute to the scissile peptide group in the substrate) in your mechanism. Use the curved arrow convention to illustrate the flow of electrons.arrow_forward
- A seven-residue peptide was found to have an amino acid composition including glu, val, Lys, met, leu, and trp (there is more than one copy of one amino acid). Several reactions on samples of the peptide resulted in the following: a) Trypsin has no effect on the heptapeptide. b) If one cycle of Edman degradation provides the PTH derivative shown below, what does this tell you about the oligopeptide under investigation? [image] c) Chymotrypsin treatment yields a dipeptide, a tetrapeptide, and a free amino acid. Possibilities? d) The tetrapeptide contains val, lys, and met (only). What information does this give you? e) Cyanogen bromide treatment generates a dipeptide, a tetrapeptide, and free Lys. f)What is the sequence of the peptide?arrow_forwardWhat would be the effect on the activity of phosphofructokinase of the mutation of Asp103 to the unusual amino acid shown below? Explain in terms of actual structures of the side chains of Asp and this unusual amino acid.arrow_forwardUnder certain conditions, peptide bond formation is more thermodynamically favorable than peptide bond hydrolysis. Would you expect chymotrypsin to catalyze peptide bond formation?arrow_forward
- UDP-glucuronosyltransferase enzymes bind the organic compound UDP-glucuronic acid (UDP-GA) in order to catalyse the transfer of a glucuronic acid group from UDP-GA to a drug molecule, releasing UDP from the active site as a product. UDP is then regenerated by the activity of another enzyme. What terms could be used to describe UDP-GA?arrow_forwardUnder certain conditions, peptide bond formation rather than peptide bond hydrolysis is thermodynamically favorable. Would you expect chymotrypsin to catalyze peptide bond formation? Explain.arrow_forwardFor the following pentapeptides: Ser-Glu-Gly-His-Ala and Gly-His-Ala-Glu-Ser Draw the most protonated forms of the pentapeptides Illustrate the ionization of the pentapeptides from the most protonated to its deprotonated form. Label the ionization with its respective pK values. Compute their isoelectric pH (pI). Do these peptides with the same amino acid composition have different net charges at pH 7.0? Explain briefly. Would you expect the titration curves of the two peptides to differ? Why or Why not?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning

Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning
Metabolic Pathways; Author: Wisc-Online;https://www.youtube.com/watch?v=m61bQYio9ys;License: Standard Youtube License