
Interpretation:
Synthesis of the given compound Darvon, starting with ethyl phenyl
Concept introduction:
Grignard reagent reactions with carbonyl groups are treated as acid–base reactions.
The reaction of Grignard reagents with carbonyl groups yields secondary and tertiary alcohols. These reactions are two-step reactions. In the first step, nucleophilic addition of carbonyl group takes place because Grignard reagent, being nucleophilic, uses its lone pair of electrons to form a bond with a carbon atom. This results in the formation of an alkoxide ion which remains associated with
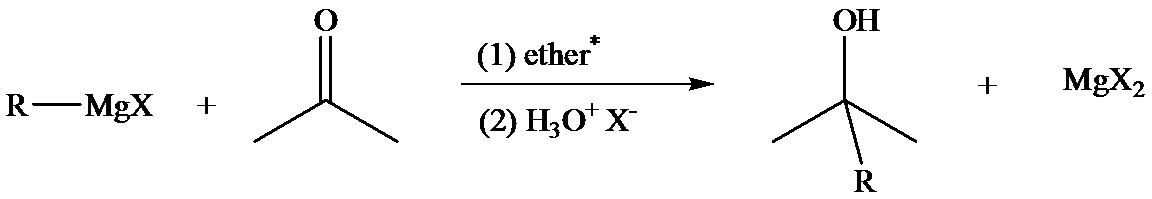
In the second step, addition of aqueous HX causes the protonation of the alkoxide ion, which leads to the formation of the alcohol and
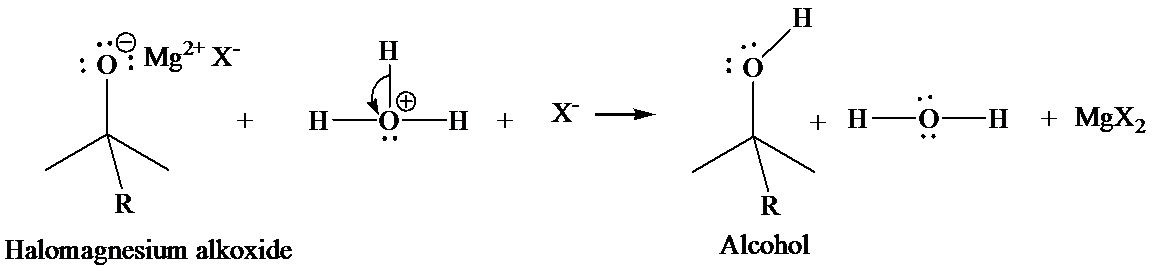
Mannich reaction: Carbonyl groups capable of forming enol react with primary or secondary
Alcohols react with acetic anhydride to form esters via nucleophilic substitution reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry
- The following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Of these three anticonvulsants, one is considerably more acidic than the other two. Which is the most acidic compound? Estimate its pKa and account for its acidity. How does its acidity compare with that of phenol? with that of acetic acid?arrow_forwardPhenylacetone can form two different enols.(a) Show the structures of these enols.(b) Predict which enol will be present in the larger concentration at equilibrium.(c) Propose mechanisms for the formation of the two enols in acid and in basearrow_forwardArrange each group of compounds in order of increasing acidity.(c) benzoic acid, o-nitrobenzoic acid, m-nitrobenzoic acidarrow_forward
- Show how you can synthesize the following compounds starting with benzene, toluene, and alcohols containing no morethan four carbon atoms as your organic starting materials. Assume that para is the major product (and separable fromortho) in ortho, para mixtures.(a) pentan-1-amine (b) N-methylbutan-1-aminearrow_forwardSuggest appropriate reagents for the following syntheses.arrow_forward3) Draw equations of the following reactions and and explain to which direction is the respective quillibrium shifted. a) cyclohexylamine + water b) aniline + sulphuric acid c) triethylamine + acetic acidarrow_forward
- The following three derivatives of succinimide are anticonvulsants that have found use in the treatment of epilepsy, particularly petit mal seizures. Q. Describe the chemistry involved in the conversion of (B) to (C). You need not present detailed mechanisms. Rather, state what is accomplished by treating (B) with NaOH and then with HCl followed by heating.arrow_forward(a) Account for the following :(i) Propanal is more reactive than propanone towards nucleophilic reagents.(ii) Electrophilic substitution in benzoic acid takes place at meta position.(iii) Carboxylic acids do not give characteristic reactions of carbonyl group.(b) Give simple chemical test to distinguish between the following pairs of compounds:(i) Acetophenone and benzaldehyde(ii) Benzoic acid and ethylbenzoate.arrow_forwardwhen acetic acid is reacted triethylamine, an acid-base reaction occurs. Write a detailed mechanisim for this reaction. Provide all reasonable resonance forms for the product.arrow_forward
- Linalool and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can't linalool be prepared by a similar pathway?arrow_forward(b) Suggest a reasonable biosynthesis for the naturally occurring alkaloid coniine (isolated from hemlock), starting from octanoic acid. Coniinearrow_forwardGive the structure of compound A.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
