
Concept explainers
Write the structures of the three products involved in this reaction sequence:
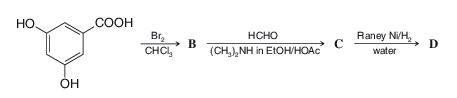
Spectral data forB:
MS(m/z): 314, 312, 310 (relative abundance 1:2:1)
1HNMR(δ): only 6.80 (s) after treatment with D2O
Data forC:
MS(m/z): 371, 369, 367 (relative abundance 1:2:1)
1HNMR(δ): 2.48 (s) and 4.99 (s) in area ratio 3:1; broad singlets at 5.5 and 11 disappeared after treatment with D2O.
Data forD:
MS(m/z):369(M+⋅−CH3) [when studied as its tris(trimethylsilyl) derivative]

1HNMR(δ): 2.16 (s) and 7.18 (s) in area ratio 3:2; broad singlets at 5.4 and 11 disappeared after treatment with D2O.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: The Central Science (13th Edition)
Fundamentals of Heat and Mass Transfer
Chemistry (7th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Inorganic Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardCompound X, shown below, spontaneously reacts via an intramolecular SN2 to produce intermediate Y, known as an "aziridinium" ion. In the presence of water, intermediate Y reacts further to form product Z. What is the identity of product Z. OH (A) + En Br O (B) aziridinium ion Y OH (C) H₂O + En Z HOarrow_forwardA task is assigned to an undergraduate student to test two samples (known as compounds K and L) in the laboratory. She placed these two compounds through various scientific tests. She discovered that these compounds have the same molecular formula, CSHSO. When treated with 2,4-dinitrophenylhydrazine, all of these compounds produce brightly coloured precipitate, and both are reduced to an organic compound with the molecular formula C§H100. However, compound K can be easily oxidized by chromic acid to formed compound N and vice versa for compound L. Furthermore, when both compounds react with Fehling's solutions, they produce negative results. However, only compound K forms a silver mirror when it reacts with Tollen's reagent, and compound L does not. Identify the possible structural formulae for compounds K, L, and N by ignoring their position isomerism. Indicate the formation of compound N from compound K. Predict the chemical reaction that occurs when compound L reacts with 2,4-…arrow_forward
- 3. cyclohexanone to cyclohexane. A second year chemistry student is assigned a task to prepare compound 2 from compound 1 using lithium aluminium hydride as a reducing agent. However, from the reaction mixture, no traces of product 2 were detected. Explain this observation in detail and suggest an alternative route that will lead to product 2. H 1 CH3 LiAllH4 H O: 2 CH3 quearrow_forwardProvide each product in the following multi-step process: (1) Br₂, hv (2) NaOCH3 (3) 9-BBN (4) H₂O₂, OH (5) PCCarrow_forward4. Complete the outlined reaction sequence by drawing structures for the major products expected. Zn(Hg) C2, 1 eq. КОН HCI(aq) UV-light heat AIC3arrow_forward
- Draw the major organic product formed in each of the following transition-metal catalyzed reactions: (a) (b) (c) (d) + H₂ + RhCI(PPH3)3 Ru(=CHPh)Cl₂(PCY3)3 CH3 Br &:0 + CH3 Br Pd(PPh 3)2Cl2 B(OH)2 Pd(OAc)2arrow_forwardThe treatment of (CH3)2C = CHCH2Br with H2O forms B (molecular formula C5H10O) as one of the products. Determine the structure of B from its H NMR and IR spectra.arrow_forwardQ1/Do as required: 1-The role of H2SO4 in Preparation of Nitrobenzene. Explain 2- Preparation of Nitrobenzene in cool phase. 3-In an experiment Hydrolysis of Acetanilide What color does the litmus paper change to and why? 4-When acetanilide reacts with bromine (x), acctanilide gives p-bromo acetanil 5- why used strongly alkaline (NaOH)in Preparation of Aniline?arrow_forward
- Provide the major product for the following reaction? (1)NBS, hv, CCI4 (2)KOH (3)arrow_forwardPossible alternative brominations include: Veratrole (1,2-dimethoxybenzene) to 1,2-dibromo-4,5-dimethoxybenzene; 4-Methylacetanilide to 2-bromo-4-methylacetanilide; 2-Methylacetanilide (made in experiment S.1) to 4-bromo-2-methylacetanilide; Vanillin to 5-bromovanillin; Acetanilide to 4-bromoacetanilide; a. b. C. d. e. EXPERIMENT S4: BROMINATION OF AROMATIC COMPOUNDS Certain other acetanilides made in experiment S.1 may also be used as precursors in this experiment. Estimated time: 1 afternoon Associated learning goals: Section 6, LG 6.6; Section 7, LG 7.2 and 7.4 Pre-lab report: complete the standard report form, and answer the following questions. In this experiment, molecular bromine (Br2) is generated from the redox reaction of potassium bromate with hydrobromic acid. Write a balanced equation for this process. Briefly outline the mechanism by which Br2 brominates your aromatic compound. Why do the bromine atoms end up at the positions indicated rather than anywhere else in the…arrow_forwardCompounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
