
Concept explainers
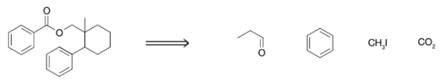
Develop a synthesis for the following molecule using only the indicated materials as sources for the carbon atoms that appear in the final product. As a hint, you will need to draw upon several reactions from earlier chapters to come up with a successful solution, and you may have to use some of the carbon sources more than one time.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry, 12e Binder Ready Version Study Guide / Student Solutions Manual
Additional Science Textbook Solutions
Organic Chemistry (8th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Organic Chemistry (9th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Chemistry: Structure and Properties (2nd Edition)
Living By Chemistry: First Edition Textbook
- A problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardFollowing is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardcomplete the following synthesesarrow_forward
- 3) Provide a synthesis of the following compounds using the given starting materials as your only carbon source and any other reagents. Br starting materials final product starting material final productarrow_forward3) A common practice that organic chemists work on is a process known as retrosynthesis. In this a chemist will take known reactants and devise a synthetic pathway to produce the desired products using reactions that they know. As you proceed through organic chemistry you will be tasked with thinking about this process. How would you make the desired final product starting from the alkyne. It will take a couple of reactions. Please show the reagents necessary. And for your last step please show the arrow pushing. Starting material Br Product Br ...arrow_forwardWrite the reagents and reaction conditions that are necessary for each of the following transformations.arrow_forward
- Provide the necessary reagents to accomplish the desired organic reactions. Please number the steps and note that some reactions will require more than one step.arrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. Draw a structural formula for the enol form of the carbonyl compound below.arrow_forwardFrom the table of reagents, select the reagents and conditions necessary to carry out the following reaction.arrow_forward
- Please give steps, intermediates, and reagents for the given synthesisarrow_forward3) Provide a synthesis of the following compounds using the given starting materials as your only carbon source and any other reagents. starting material final productarrow_forwardByphenyl synthesis. From benzene, organic compounds with 2 carbons or less, and any other inorganic compound you want. Synthesize the following molecule.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

