
Fundamentals of Physics Extended
10th Edition
ISBN: 9781118230725
Author: David Halliday, Robert Resnick, Jearl Walker
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 7Q
(a) Rank the four paths of Fig. 19-16 according to the work done by the gas, greatest first, (b) Rank paths 1, 2, and 3 according to the change in the internal energy of the gas, most positive first and most negative last.
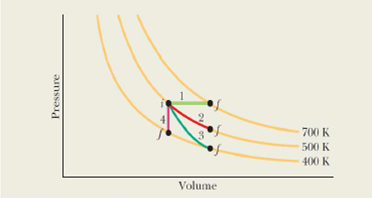
Figure 19-16 A p-V diagram representing four special process for an ideal monatomic gas.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Fundamentals of Physics Extended
Ch. 19 - For four situations for an ideal gas, the table...Ch. 19 - In the p-V diagram of Fig. 19-17, the gas does 5 J...Ch. 19 - For a temperature increase of T1, a certain amount...Ch. 19 - The dot in Fig, 19-18a represents the initial...Ch. 19 - A certain amount of energy is to be transferred as...Ch. 19 - The dot in Fig. 19-18b represents the initial...Ch. 19 - a Rank the four paths of Fig. 19-16 according to...Ch. 19 - The dot in Fig. 19-18c represents the initial...Ch. 19 - Prob. 9QCh. 19 - Does the temperature of an ideal gas increase,...
Ch. 19 - Prob. 1PCh. 19 - Gold has a molar mass of 197 g/mol. a How many...Ch. 19 - SSM Oxygen gas having a volume of 1000 cm3 at...Ch. 19 - A quantity of ideal gas at: 10.0C and 100 kPa...Ch. 19 - The best laboratory vacuum has a pressure of about...Ch. 19 - Water bottle in a hot car. In the American...Ch. 19 - Suppose 1.80 mol of an ideal gas is taken from a...Ch. 19 - Compute a the number of moles and b the number of...Ch. 19 - An automobile tire has a volume of 1.64 102 m3...Ch. 19 - A container encloses 2 mol of an ideal gas that...Ch. 19 - SSM ILW WWW Air that initially occupies 0.140 m3...Ch. 19 - GO Submarine rescue. When the U.S. submarine...Ch. 19 - Prob. 13PCh. 19 - In the temperature range 310 K to 330 K, the...Ch. 19 - Suppose 0.825 mol of an ideal gas undergoes an...Ch. 19 - An air bubble of volume 20 cm3 is at the bottom of...Ch. 19 - GO Container A in Fig. 19-22 holds an ideal gas at...Ch. 19 - The temperature and pressure in the Suns...Ch. 19 - a Compute the rms speed of a nitrogen molecule at...Ch. 19 - Calculate the rms speed of helium atoms at 1000 K....Ch. 19 - SSM The lowest possible temperature in outer space...Ch. 19 - Find the rms speed of argon atoms at 313 K. See...Ch. 19 - A beam of hydrogen molecules H2 is directed toward...Ch. 19 - At 273 K and 1.00 102 atm, the density of a gas...Ch. 19 - Prob. 25PCh. 19 - Prob. 26PCh. 19 - Water standing in the open at 32.0C evaporates...Ch. 19 - At what frequency would the wavelength of sound in...Ch. 19 - SSM The atmospheric density at an altitude of 2500...Ch. 19 - Prob. 30PCh. 19 - In a certain particle accelerator, protons travel...Ch. 19 - Prob. 32PCh. 19 - Prob. 33PCh. 19 - Prob. 34PCh. 19 - Prob. 35PCh. 19 - The most probable speed of the molecules in a gas...Ch. 19 - Prob. 37PCh. 19 - Figure 19-24 gives the probability distribution...Ch. 19 - At what temperature does the rms speed of a...Ch. 19 - Two containers are at the same temperature. The...Ch. 19 - Prob. 41PCh. 19 - What is the internal energy of 1.0 mol of an ideal...Ch. 19 - Prob. 43PCh. 19 - GO One mole of ail ideal diatomic gas goes from a...Ch. 19 - ILW The mass of a gas molecule can be computed...Ch. 19 - Under constant pressure, the temperature of 2.00...Ch. 19 - The temperature of 2.00 mol of an ideal monatomic...Ch. 19 - GO When 20.9 J was added as heat to a particular...Ch. 19 - SSM A container holds a mixture of three...Ch. 19 - We give 70 J as heat to a diatomic gas, which then...Ch. 19 - Prob. 51PCh. 19 - GO Suppose 12.0 g of oxygen O2 gas is heated at...Ch. 19 - SSM WWW Suppose 4.00 mol of an ideal diatomic gas...Ch. 19 - We know that for an adiabatic process pV = a...Ch. 19 - A certain gas occupies a volume of 4.3 L at a...Ch. 19 - Suppose 1.00 L of a gas with = 1.30, initially at...Ch. 19 - The volume of an ideal gas is adiabatically...Ch. 19 - GO Opening champagne. In a bottle of champagne,...Ch. 19 - GO Figure 19-26 shows two paths that may be taken...Ch. 19 - GO Adiabatic wind. The normal airflow over the...Ch. 19 - GO A gas is to be expanded from initial state i to...Ch. 19 - GO An ideal diatomic gas, with rotation but no...Ch. 19 - Figure 19-27 shows a cycle undergone by 1.00 mol...Ch. 19 - Calculate the work done by an external agent...Ch. 19 - An ideal gas undergoes an adiabatic compression...Ch. 19 - Prob. 66PCh. 19 - An ideal monatomic gas initially has a temperature...Ch. 19 - Prob. 68PCh. 19 - SSM The envelope and basket of a hot-air balloon...Ch. 19 - An ideal gas, at initial temperature T1 and...Ch. 19 - Prob. 71PCh. 19 - At what temperature do atoms of helium gas have...Ch. 19 - Prob. 73PCh. 19 - Prob. 74PCh. 19 - The temperature of 3.00 mol of a gas with CV =...Ch. 19 - During a compression at a constant pressure of 250...Ch. 19 - SSM Figure 19-28 shows a hypothetical speed...Ch. 19 - Prob. 78PCh. 19 - SSM An ideal gas undergoes isothermal compression...Ch. 19 - Oxygen O2 gas at 273 K and 1.0 atm is confined to...Ch. 19 - An ideal pas is taken through a complete cycle in...Ch. 19 - Prob. 82PCh. 19 - SSM A sample of ideal gas expands from an initial...Ch. 19 - An ideal gas with 3.00 mol is initially in state 1...Ch. 19 - A steel lank contains 300 g of ammonia gas NH3 at...Ch. 19 - In an industrial process the volume of 25.0 mol of...Ch. 19 - Figure 19-29 shows a cycle consisting of five...Ch. 19 - An ideal gas initially at 300 K is compressed at a...Ch. 19 - A pipe of length L = 25.0 m that is open at one...Ch. 19 - In a motorcycle engine, a piston is forced down...Ch. 19 - For adiabatic processes in an ideal gas, show that...Ch. 19 - Air at 0.000C and 1.00 atm pressure has a density...Ch. 19 - Prob. 93PCh. 19 - Prob. 94PCh. 19 - Prob. 95PCh. 19 - For air near 0C, by how much does the speed of...Ch. 19 - Prob. 97P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Theorists have had spectacular success in predicting previously unknown particles. Considering past theoretical...
College Physics
16. (II) A golf ball of mass 0.045 kg is hit off the lee at a speed of 38 m/s. The golf club was in contact wit...
Physics: Principles with Applications
An aluminum calorimeter with a mass of 100 g contains 250 g of water. The calorimeter and water are in thermal ...
Physics for Scientists and Engineers
6. A helium-neon laser beam has a wavelength in air of 633 nm. It takes 1.38 ns for the light to travel through...
College Physics: A Strategic Approach (3rd Edition)
34. A particle has a charge of −3.00 nC. (a) Find the magnitude and direction of the electric field due to this...
College Physics (10th Edition)
A thermometer that indicates the temperature with a liquid column has a bulb with a larger volume of liquid. Wh...
Fundamentals Of Thermodynamics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- For a temperature increase of 10 at constant volume, what is the heat absorbed by (a) 3.0 mol of a dilute monatomic gas; (b) 0.50 mol of a dilute diatomic gas; and (c) 15 mol of a dilute polyatomic gas?arrow_forwardAn ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forward
- One mole of an ideal gas does 3 000 J of work on its surroundings as it expands isothermally to a final pressure of 1.00 atm and volume of 25.0 L. Determine (a) the initial volume and (b) the temperature of the gas.arrow_forwardAn ideal gas has a pressure of 0.50 atm and a volume of 10 L. It is compressed adiabatically and quasi-statically until its pressure is 3.0 atm and its volume is 2.8 L. Is the monatomic, diatomic, or polyatomic?arrow_forwardDuring the power stroke in a four-stroke automobile engine, the piston is forced down as the mixture of combustion products and air undergoes an adiabatic expansion. Assume (1) the engine is running at 2 500 cycles/min; (2) the gauge pressure immediately before the expansion is 20.0 atm; (3) the volumes of the mixture immediately before and after the expansion are 50.0 cm3 and 400 cm3, respectively (Fig. P21.31); (4) the time interval for the expansion is one-fourth that of the total cycle; and (5) the mixture behaves like an ideal gas with specific heat ratio 1.40. Find the average power generated during the power stroke.arrow_forward
- A gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path 1.8pV= constant. (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.arrow_forwardAn amount of n moles of a monatomic ideal gas in a conducting container with a movable piston is placed in a large thermal heat bath at temperature T1 and the gas is allowed to come to equilibrium. After the equilibrium is leached, the pressure on the piston is lowered so that the gas expands at constant temperature. The process is continued quasi-statically until the final pressure is 4/3 of the initial pressure p1 . (a) Find the change in the internal energy of the gas. (b) Find the work done by the gas. (c) Find the heat exchanged by the gas, and indicate, whether the gas takes in or gives up heat.arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forward
- An aluminum rod 0.500 m in length and with a cross-sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 300 K. (a) If one-half of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool. (b) If the circular surface of the upper end of the rod is maintained at 300 K, what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 W/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forwardTwo moles of a monatomic ideal gas such as helium is compressed adiabatically and reversibly from a state (3 atm, 5 L) to a state with pressure 4 atm. (a) Find the volume and temperature of the final state. (b) Find the temperature of the initial state of the gas. (c) Find the work done by the gas in the process. (d) Find the change in internal energy of the gas in the process.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY