
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
9th Edition
ISBN: 9781337034623
Author: McMurry
Publisher: CENGAGE C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1.SE, Problem 57AP
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol).
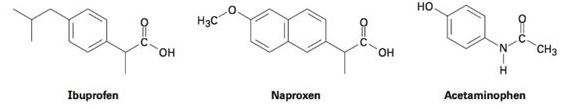
(a) How many sp3-hybridized carbons does each molecule have?
(b) How many sp2-hybridized carbons does each molecule have?
(c) Can you spot any similarities in their structures?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such as ibuprofen. How many sp3-hybridized carbons and sp2-hybridized carbons does the molecule have?
The N atom in CH3CONH2 (acetamide) is sp2 hybridized, even though it is surrounded by four groups. Using this information, draw a diagram that shows the orbitals used by the atoms in the – CONH2 portion of acetamide, and offer an explanation as to the observed hybridization.
does this compound contain an sp2-hybridized carbon atom?
Chapter 1 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
Ch. 1.3 - Give the ground-state electron configuration for...Ch. 1.3 - How many electrons does each of the following...Ch. 1.4 - Prob. 3PCh. 1.4 - Convert the following representation of ethane,...Ch. 1.4 - What are likely formulas for the following...Ch. 1.4 - Prob. 6PCh. 1.4 - Prob. 7PCh. 1.7 - Draw a line-bond structure for propane, CH3CH2CH3....Ch. 1.7 - Convert the following molecular model of hexane, a...Ch. 1.8 - Draw a line-bond structure for propene, CH3CH=CH2....
Ch. 1.8 - Draw a line-bond structure for 1, 3-butadiene,...Ch. 1.8 - Following is a molecular model of aspirin...Ch. 1.9 - Draw a line-bond structure for propyne, CH3C≡CH....Ch. 1.10 - Prob. 14PCh. 1.12 - Prob. 15PCh. 1.12 - Prob. 16PCh. 1.12 - The following molecular model is a representation...Ch. 1.SE - Convert each of the following molecular models...Ch. 1.SE - The following model is a representation of citric...Ch. 1.SE - The following model is a representation of...Ch. 1.SE - The following model is a representation of...Ch. 1.SE - How many valence electrons does each of the...Ch. 1.SE - Give the ground-state electron configuration for...Ch. 1.SE - Prob. 24APCh. 1.SE - Prob. 25APCh. 1.SE - Draw an electron-dot structure for acetonitrile,...Ch. 1.SE - Draw a line-bond structure for vinyl chloride,...Ch. 1.SE - Fill in any nonbonding valence electrons that are...Ch. 1.SE - Convert the following line-bond structures into...Ch. 1.SE - Convert the following molecular formulas into...Ch. 1.SE - Prob. 31APCh. 1.SE - Oxaloacetic acid, an important intermediate in...Ch. 1.SE - Prob. 33APCh. 1.SE - Potassium methoxide, KOCH3, contains both covalent...Ch. 1.SE - What is the hybridization of each carbon atom in...Ch. 1.SE - Prob. 36APCh. 1.SE - Prob. 37APCh. 1.SE - What bond angles do you expect for each of the...Ch. 1.SE - Propose structures for molecules that meet the...Ch. 1.SE - What kind of hybridization do you expect for each...Ch. 1.SE - Pyridoxal phosphate, a close relative of vitamin...Ch. 1.SE - Prob. 42APCh. 1.SE - Prob. 43APCh. 1.SE - Quetiapine, marketed as Seroquel, is a heavily...Ch. 1.SE - Tell the number of hydrogens bonded to each carbon...Ch. 1.SE - Why do you suppose no one has ever been able to...Ch. 1.SE - Allene, H2C=C=CH2, is somewhat unusual in that it...Ch. 1.SE - Allene (see Problem 1-47) is structurally related...Ch. 1.SE - Complete the electron-dot structure of caffeine,...Ch. 1.SE - Most stable organic species have tetravalent...Ch. 1.SE - A carbanion is a species that contains a...Ch. 1.SE - Divalent carbon species called carbenes are...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - Prob. 56APCh. 1.SE - Among the most common over-the-counter drugs you...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Glyphosate (below) is a common herbicide that is relatively harmless to animals but deadly to most plants. Describe the shape around and the hybridization of the P, N, and three num-bered C atoms.arrow_forward(a) Determine the hybridization of each N atom in ibrutinib, a drug used to treat mantle cell lymphoma and chronic lymphocytic leukemia. (b) In what type of orbital does the lone pair on each N atom reside? (c) How many sp2 hybridized atoms does ibrutinib contain?arrow_forwardHow many unhybridized P Porter orbitals are there in an sp2 hybridized C atom and in an sp2 hybridized O atom? A. 1 each B. 2 each C. 2 in the C atom and 1 in the O D. 1 in the C atom and 2 in the Oarrow_forward
- borazine B3N3H6 is a cyclic compound with alternatig B and N atoms and is isoeletronic with benzene. Identify the hybridization of the N and B atoms. Are there any delocalized electrons in borazine?arrow_forwardDetermine whether each structure is likely to be colored or not. If colored, indicating the extented conjugation by marking series of continuous sp2 hybridized atomsarrow_forwardHow many sp2 hybridized atoms does ibrutinib contain?arrow_forward
- Following is a molecular model of aspirin (acetylsalicylic acid). Identify the hybridization of the orbitals on each carbon atom in aspirin, and tell which atoms have lone pairs of electrons (gray = C, red = O, ivory = H).arrow_forwardIdentify all the central atoms that are sp3d2-hybridized in the following molecule:arrow_forwardHow many carbons can be described as sp2 hybridized in this molecule? H3C---CH=CH---CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY