
Pearson eText for Biochemistry: Concepts and Connections -- Instant Access (Pearson+)
2nd Edition
ISBN: 9780137533114
Author: Dean Appling, Spencer Anthony-Cahill
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 18P
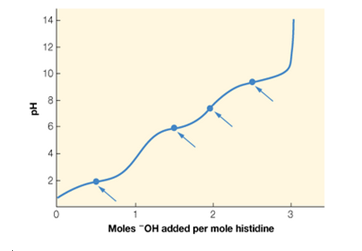
Histidine is an amino acid with three titratable groups: an -NH3+ group (pKa = 9.2) —COOH group (pKa = 1.8). and an imidazole (
a. Identify which point on the titration curve corresponds to the pKa for each of the titratable groups, and which point corresponds to the pl. Explain your choices.
b. Calculate the value of pl for histidine.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
E. Histidine is an amino acid with three titratable groups: an -NH," group
(pK, = 9.2), a -COOH group (pK, = 1.8), and an imidazole (amine-
like) group (pK, = 6.0). The titration curve for histidine is shown below
with four points highlighted.
(a) Identify which point on the titration curve corresponds to the pK, for
each of the titratable groups, and which point corresponds to the pl.
Explain your choices.
(b) Calculate the value of pl for histidine.
14
12
10
6.
4
2-
2
3
Moles "OH added per mole histidine
на
LEFT:You have the following titration curve of an amino acid with a non-polar R-group with
arrows on curve from (1) to (5).
RIGHT: This amino acid has the following structures (a) to (e)) at different points of the
titration curve. Match each numbered arrow on the curve (from (1) to (5)) with the appropriate
amino acid structure (from (a) to (e))
12
(5)
H.NCHCOH + HNCHCO
(a)
10
(4)
CH3
CH
Isoelectrin
HŃCHCO +
(b)
ppint
43)
HNCHCO
CH3
CH3
(2)
HaNCHCOH
H. NCHCO (e)
(d
CH3
CH3
0.0
0.5
1.0
15
20
NaOH equivalents.
H-NCHCO
(e)
CH3
LEFT You have the following titration curve of an amino acid with a non-polar R-group with
arrows on curve from (1) to (5).
RIGHT. This amino acid has the following structures ((a) to (e)) at different points of the
titration curve. Match each numbered arrow on the curve (from (1) to (5) with the appropriate
amino acid structure (from (a) to (e))
12
(5)
H,NCHCOH + H NCHC
(a)
10
(4)
CH,
CH
Isoelectrie
HNCHCO+
(b)
point
HNCHCO
43)
CH
CH
(2)
HNCHCOH
H,NCHCO (C)
CH3
CH3
0.0
0.5
1.5
20
NaOH equivalents
HNCHCO
(e)
CH3
Chapter 2 Solutions
Pearson eText for Biochemistry: Concepts and Connections -- Instant Access (Pearson+)
Ch. 2 - Suppose a chloride ion and a sodium ion are...Ch. 2 - Draw two different possible hydrogen-bonding...Ch. 2 - Prob. 3PCh. 2 - 4. What is the pH of each of the following...Ch. 2 - Prob. 5PCh. 2 - The weak acid HA is 2% ionized (dissociated) in a...Ch. 2 - 7. Calculate the pH values and draw the titration...Ch. 2 - What is the pH of the following buffer mixtures?...Ch. 2 - a. Suppose you wanted to make a buffer of exactly...Ch. 2 - Prob. 10P
Ch. 2 - You need to make a buffer whose pH is 7.0, and you...Ch. 2 - Describe the preparation of 2.00 L of 100 glycine...Ch. 2 - Carbon dioxide is dissolved in blood (pH 7.4) to...Ch. 2 - What is the molecular basis for the observation...Ch. 2 - The anno acid arginine ionizes according to the...Ch. 2 - It is possible to make a buffer that functions...Ch. 2 - A student is carrying out a biological preparation...Ch. 2 - Histidine is an amino acid with three titratable...Ch. 2 - Prob. 19PCh. 2 - A biochemical reaction takes place in a 1.00 ml...Ch. 2 - Is RNA-binding enzyme RNase A more likely to have...Ch. 2 - Consider a protein in which a negatively charged...Ch. 2 - Prob. 23PCh. 2 - Prob. 24P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Write a structural formula for a tetrapeptide composed of phenyl-alanine, glycine, aspartic acid, and histidine in which the phenylalanine is N-terminal and the histidine is C-terminal. How many different tetrapeptides could be written that fit the criteria in this question?arrow_forwardFor the following pentapeptides: Ser-Glu-Gly-His-Ala and Gly-His-Ala-Glu-Ser A. Compute their isoelectric pH (pI). Show full solution. Use standard pKa values. B. Do these peptides with the same amino acid composition have different net charges at pH 7.0? Explain briefly. C. Would you expect the titration curves of the two peptides to differ? Why or Why not?arrow_forwardThe melanocyte-stimulating peptide hormone a-melanotropin has the following sequence: Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val (a) Write the sequence using the one-letter abbreviations. (b) Calculate the molecular weight of a-melanotropin, a da'arrow_forward
- Polylysine adopts a random structure in solution at physiological pH (i.e. 7.4). Given that the e-amino group of lysine has a pKa of 10.5, under what circumstances do you think polylysine will form an a-helix? Give an explanation for your decision. Given the pKa of the side-chain COOH group, under what circumstances would you anticipate polyglutamate to form an a-helix?arrow_forwardZ-scale descriptors are also calculated for artificial (synthetic) amino acids. Which of the four following descriptions could correspond to amino-phenylacetate and which to homoarginine? Justify your answer! 1) z1=-3.51 z2=2.93 z3=2.94 2) z1=2.70 z2=3.06 z3=-4.15 3) z1=-3.51 z2=-2.93 z3=2.94 4) z1=2.70 z2=-3.06 z3=arrow_forwardThe bacterially produced antibiotic gramicidin A forms channels in cell membranes that allow the free diff usion of Na+ and K+ ions, thereby killing the cell. This peptide consists of a sequence of Dand L-amino acids. The sequence of a segment of fi ve amino acids in gramicidin A is R-Gly-L-Ala-D-Leu-L-Ala-D-Val-R′. Complete the Fischer projection below by adding the correct group to each vertical bond.arrow_forward
- Complete the following table by providing the Fischer and Haworth Projections of the given sugars. (Please be careful with the alpha and beta, and the complete and abbreviated) (not a graded question) Sugar Fischer Projection Haworth Projection (Complete) alpha-anomer Haworth Projection (Abbreviated) beta-anomer L-Galactose L-Fructosearrow_forwardThe melanocyte-stimulating peptide hormone α-melanotropin has the following sequence: Ser–Tyr–Ser–Met–Glu–His–Phe–Arg–Trp–Gly–Lys–Pro–Val(a) Write the sequence using the one-letter abbreviations.(b) Calculate the molecular weight of α-melanotropin, using data in as shown.arrow_forwardHow many different tripeptides can be formed from three different amino acids, glutamine, histidine, and lysine? Draw the possible structures. Using three-letter abbreviations, give the names for all of the possible tripeptides.arrow_forward
- What would the following sequence of dodecapeptide M be? My best guess would be option Carrow_forwardFollowing are structural formulas for cytosine and thymine. Draw two additional tautomeric forms for cytosine and three additional tautomeric forms for thymine.arrow_forwardThere are two parts to this problem. A) the pl for Histidine using the titration curve. Choices: 4 6 8 10 12 B) pKA value for Histidine usind the titration curve. Choices, can be more than 1: 1 2 4 6 8 10 12 Thank You!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY