
Explain why each is an incorrect IUPAC name and write the correct IUPAC name for the intended compound.
- (a) 1,3-Dimethylbutane
- (b) 4-Methylpentane
- (c) 2,2-Diethylbutane
- (d) 2-Ethyl-3-methylpentane
- (e) 2-Propylpentane
- (f) 2,2-Diethylheptane
- (g) 2,2-Dimethylcyclopropane
- (h) 1-Ethyl-5-methylcyclohexane
(a)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
1,3-Dimethylbutane
Correct IUPAC name:
The structure of 1,3-Dimethylbutane is,
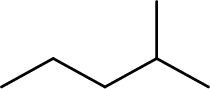
The given IUPAC name is incorrect because in the above structure, the longest chain is pentane.
The main carbon chain has five carbon atoms and one methyl group at the second position
Hence, the correct IUPAC name is 2-methylpentane.
(b)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
4-Methylpentane
Correct IUPAC name:
The structure of 4-Methylpentane is,
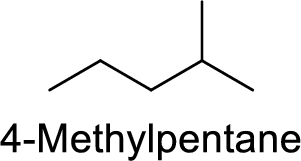
The given IUPAC name is incorrect because in the above structure, the pentane is numbered wrongly.
The main carbon chain has five carbon atoms and one methyl group at the second position
Hence, the correct IUPAC name is 2-methylpentane.
(c)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
2,2-Diethylbutane
Correct IUPAC name:
The structure of 2,2-Diethylbutane is,
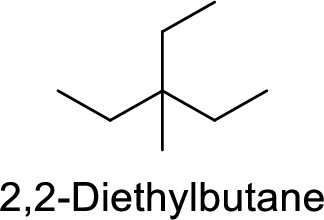
The given IUPAC name is incorrect because in the above structure, the longest chain is pentane.
The main carbon chain has five carbon atoms and one methyl group and one ethyl group at the third position
Hence, the correct IUPAC name is 3-ethyl-3-methylpentane.
(d)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
2-Ethyl-3-methylpentane
Correct IUPAC name:
The structure of 2-Ethyl-3-methylpentane is,
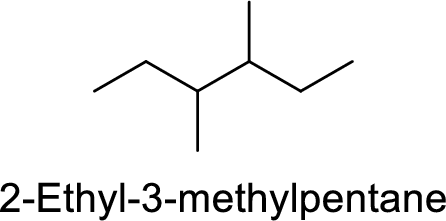
The given IUPAC name is incorrect because in the above structure, the longest chain is hexane.
The main carbon chain has six carbon atoms and two methyl group at the third and fourth position
Hence, the correct IUPAC name is 3,4-dimethylhexane.
(e)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituents must be chosen as parent chain.
Explanation of Solution
Given data:
2-Propylpentane
Correct IUPAC name:
The structure of 2-Propylpentane is,
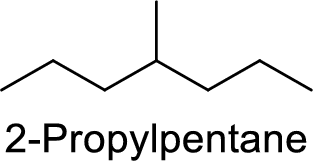
The given IUPAC name is incorrect because in the above structure, the longest chain is heptane.
The main carbon chain has seven carbon atoms and one methyl group at the fourth position
Hence, the correct IUPAC name is 4-methylheptane.
(f)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
2,2-Diethylheptane
Correct IUPAC name:
The structure of 2,2-Diethylheptane is,
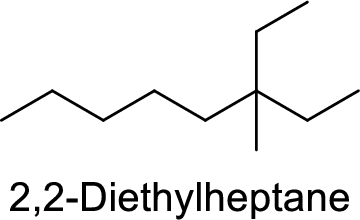
The given IUPAC name is incorrect because in the above structure, the longest chain is octane.
The main carbon chain has eight carbon atoms and one methyl group and one ethyl group are at the second carbon.
Hence, the correct IUPAC name is 3-ethyl-3-methyloctane.
(g)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
2,2-Dimethylcyclopropane
Correct IUPAC name:
The structure of 2,2-Dimethylcyclopropane is,
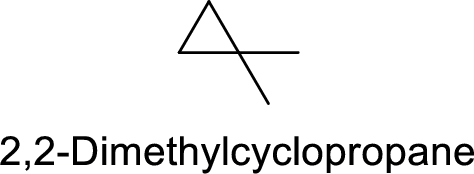
The ring is numbered incorrectly for the above structure.
The main core of the given compound has a cyclic three membered ring with two methyl groups at one carbon.
Hence, the correct IUPAC name is 1,1-dimethylcylcopropane.
(h)
Interpretation:
The reason for the incorrect IUPAC name of the given compound has to be explained also the correct IUPAC name for the intended compound has to be given.
Concept Introduction:
IUPAC nomenclature:
The rules for writing IUPAC system of names is as follows,
- 1) For an alkane with unbranched chain of carbon atoms, prefix the root word showing the number of carbon atoms ad end the name with –ane.
- 2) For branched chain alkane, select the longest chain of carbon atoms as the parent chain, its name becomes the root name.
- 3) Number all the carbon atoms in the parent chain. When a substituent is attached to the chain, the number shows the carbon atom to which the substituent is attached. Use a hyphen in the name to connect the number to the substituent name.
- 4) Always number the parent chain in such a way that the carbon bearing substituent gets the lower number.
- 5) If a same substituent is present more than one time, then number the parent chain in such a way that the first encountered substituent gets the lowest number. Prefix di-, tri-, tetra-, penta- and so on to indicate the number of times the substituent occurs. A comma is used to separate position numbers.
- 6) If there are two or more different substituents, list them in alphabetical order and number the chain from the end that gives the lower number to the substituent encountered first. If there are different substituents in equivalent positions on opposite ends of the parent chain, give the substituent of lower alphabetical order the lower number.
- 7) Alphabetize the names of the substituents first and then insert these prefixes.
- 8) Where two or more parent chains of identical lengths are present, the chain with greater number of substituent must be chosen as parent chain.
Explanation of Solution
Given data:
1-Ethyl-5-methylcyclohexane
Correct IUPAC name:
The structure of 1-Ethyl-5-methylcyclohexane is,
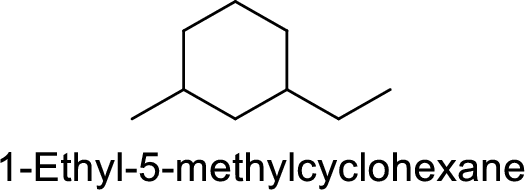
The main core of the given compound has a cyclic six membered ring with one ethyl groups at first carbon and one methyl group at third carbon.
The main carbon chain has six carbon atoms and one methyl group at the second position
Hence, the correct IUPAC name is 1-ethyl-3-methylcyclohexane.
Want to see more full solutions like this?
Chapter 2 Solutions
ORG.CHEM:TXT+OWLV2+MINDTAP 6MTHS >BI<
- Write a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forwardDraw the structures and give the name of the 8 constitutional isomers with the molecular formula C9H12 that contain a benzene ring.arrow_forwardPlease help for all questions A) Convert the structure in condensed form.b) Identify one the functional group with its respective homologous series that exists in thecompound.c) Draw one possible structure with molecular formula C6H12 that has two tertiary carbon.d) Arrange the following compounds in increasing order of melting point.Hexane 3-methylhexane 3-ethylhexane 2,3-dimethylpentanearrow_forward
- C5H8Cl2 can you name and draw all isomers , make sure each has a 5 membered cyclic ring . And use E,Z when approparrow_forwardName all unbranched ether and alcohol isomers with formula C5H12O, and write their structural formulas.arrow_forwardMenthene, a compound found in mint plants, has the formula C10H18 and the IUPAC name 1-isopropyl- 4-methylcyclohexene. What is the structure of menthene?arrow_forward
- Draw all possible structure(s) and give the IUPAC systematic name(s) of an alkane or cycloalkane with the formula C8H18 that has only primary hydrogen atoms...arrow_forwardDraw a structural diagram for 1-pentanol. - you do not have to consider stereochemistry - you do not have to explicitly draw H atoms - CH4 is given to youarrow_forwardWhy (a) is cis 3,4dimethylhex2ene? Why (b) is trans 6 Methylhept3ene?arrow_forward
- Draw the structures and give the name of the 8 constitutional isomers withe the molecular formula C9H12 that contain a benzene ring.arrow_forwardAll of the following names are incorrect or incomplete. In each case, draw the structure (or a possible structure) and name it correctly.(i) 2-bromo-3-ethylbutane (ii) 2,3-diethyl-5-isopropylheptanearrow_forward(5c-102) Draw “4-ethyl-3-methylpentane.” Is this the correct IUPAC name for this structure? If not, what should the molecule be named, according to IUPAC rules?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





