
(a)
Interpretation:
Symbol of atom or ion has to be written that has 23 protons, 28 neutrons, and 20 electrons.
Concept Introduction:
Each and every element present in the Periodic table has a unique name. Some of the elements are named considering their
Chemical names are represented as atomic symbols. In the symbols, the mass number and atomic number are shown. The complete
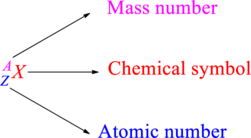
Atomic number is the total number of protons present in the atom of an element. Mass number is the total number of protons and neutrons present in nucleus of an atom.
(a)
Explanation of Solution
The species is said to contain 23 protons, 28 neutrons, and 20 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 23 is vanadium. The atomic symbol of ion can be given as
(b)
Interpretation:
Symbol of atom or ion has to be written that has 53 protons, 74 neutrons, and 54 electrons.
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
The species is said to contain 53 protons, 74 neutrons, and 54 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 53 is iodine. The atomic symbol of ion can be given as
(c)
Interpretation:
Symbol of atom or ion has to be written that has 44 protons, 58 neutrons, and 41 electrons.
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
The species is said to contain 44 protons, 58 neutrons, and 41 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
Element with the atomic number of 44 is ruthenium. The atomic symbol of ion can be given as
(d)
Interpretation:
Symbol of atom or ion has to be written that has 15 protons, 16 neutrons, and 15 electrons.
Concept Introduction:
Refer part (a).
(d)
Explanation of Solution
The species is said to contain 15 protons, 16 neutrons, and 15 electrons. Atomic number is equal to the number of protons and mass number is equal to the sum of protons and neutrons. Atomic number and mass number can be given as shown below.
The charge on the ion can be calculated considering the number of protons and electrons as shown below.
As the charge is zero, this is an atom and not an ion.
Element with the atomic number of 15 is phosphorus. The atomic symbol of atom can be given as
Want to see more full solutions like this?
Chapter 2 Solutions
Chemistry: Principles and Practice
- What are the live most abundant elements (by mass) in the earth’s crust, oceans, and atmosphere?arrow_forwardhe vigorous reaction between aluminum and iodine gives the balanced equation: :math>2Al(s)+3I2(s)2AlI2(s). mg src=Images/HTML_99425-9-2QAP_image001.jpg alt="" align="top"/> at do the coefficients in this balanced chemical equation tell us about the proportions in which these substances react on a macroscopic (mole) basis?arrow_forwardThe element bromine is Br2, so the mass of a Br2 molecule is the sum of the mass of its two atoms. Bromine has two isotopes. The mass spectrum of Br2 produces three peaks with relative masses of 157.836, 159.834, and 161.832, and relative heights of 6.337, 12.499. and 6.164, respectively. (a) What isotopes of bromine are present in each of the three peaks? (b) What is the mass of each bromine isotope? (c) What is the average atomic mass of bromine? (d) What is the abundance of each of the two bromine isotopes?arrow_forward
- If mass Q/ mass A in AQ= 0.271 and mass Q/mass A in AxQy= 0.362, what is the formual of AxQy?arrow_forwardWhat would be the mass, in kilograms, of a block of graphite consisting of 1.55 x 1026 carbon atoms?arrow_forwardA new compound has the empirical formula GaCl2. Thissurprises some chemists who, based on the position of gallium in the periodic table, expect a chloride of gallium to have the formula GaCl3 or possibly GaCl. They suggest that the “GaCl2” is really Ga[GaCl4], in which the bracketed group behaves as a unit with a -1 charge. Suggest experiments to test this hypothesisarrow_forward
- Boron, atomic number 5, occurs naturally as two isotopes, 10B and 11B, with natural abundances of 19.9% and 80.1%, respectively. (a) Will the mass percentage of F be the same in 10BF3 and 11BF3? If not, why is that the case?arrow_forwardAlthough carbon-12 is now used as the standard for atomic weights, this has not always been the case. Early attempts at classification used hydrogen as the standard, with the weight of hydrogen being set equal to 1.0000. Later attempts defined atomic weights using oxygen (with a weight of 16.0000 ). In each instance, the atomic weights of the other elements were defined relative to these masses. (To answer this question, you need more precise data on current atomic weights: H,1.00794 u; O, 15.9994 u.) (a) If H=1.0000 u was used as a standard for atomic weights, what would the atomic weight of oxygen be? What would be the value of Avogadro's number under these circumstances?(b) Assuming the standard is O=16.0000 u, determine the value for the atomic weight of hydrogen and the value of Avogadro's number.arrow_forwardDescribe and distinguish between thelaw of conservation of mass, the law ofconstant composition, and the law ofmultiple proportions.arrow_forward
- Given that you had 1 mol glucose (C 6H 12O 6), what would the mole ratio of glucose to oxygen be?arrow_forwardWhat is the mass, in grams, of Vitamin C (C6H8O6) that contains 1.076 grams of carbon?arrow_forward(a) Assuming the dimensions of the nucleus and atomshown in Figure 2.10, what fraction of the volume of the atomis taken up by the nucleus? (b) Using the mass of the protonfrom Table 2.1 and assuming its diameter is 1.0 x 10-15 m,calculate the density of a proton in g/cm3.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



