
Concept explainers
(a)
Interpretation:
The ground state electronic configuration of silicon has to be given.
Concept Introduction:
The distribution of electrons in various orbitals is known as electronic configuration.
The filling of orbitals in the ground state is determined by the following rule:
Aufbau Principle:
The Aufbau principle states that in the ground state of an atom the orbital with lower energy are filled up first before the filling of the orbital with ah higher energy commences.
(b)
Interpretation:
The valence electrons of silicon have to be given.
Concept Introduction:
Valence electrons:
Valence electrons are electrons that exist in the outermost shell surrounding an atomic nucleus.
(c)
Interpretation:
The electron dot structure of silicon has to be given.
Concept Introduction:
Electron dot structure:
A Lewis dot diagram is a representation of the valence electrons of an atom that uses dots around the
Example:
The valence electron of Mg is two it can be represented in electron dot structure as,
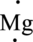
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Using Lewis electron-dot symbols to depict the monatomic ions formed from each of the following reactants, predict the formula of the compound the ions produce.(Type your answer using the format CO2 for CO2.) (a) O and Ca (b) N and Mg (c) Br and Li (d) K and Parrow_forward(a) Would you expect scandium oxide to be a solid, liquid, or gas at room temperature?arrow_forwardBoron, atomic number 5, occurs naturally as two isotopes, 10B and 11B, with natural abundances of 19.9% and 80.1%, respectively. (a) In what ways do the two isotopes differ from each other? Does the electronic configuration of 10B differ from that of 11B? (b) Draw the orbital diagram for an atom of 11B. Which electrons are the valence electrons? (c) Indicate three ways in which the 1s electrons in boron differ from its 2s electrons. (d) Elemental boron reacts with fluorine to form BF3, a gas. Write a balanced chemical equation for the reaction of solid boron with fluorine gas. (e) ΔHf° for BF3(g) is -1135.6 kj/mol. Calculate the standard enthalpy change in the reaction of boron with fluorine. (f) Will the mass percentage of F be the same in 10BF3 and 11BF3? If not, why is that the case?arrow_forward
- Arrange in order of increasing nonmetallic character. (Use the appropriate <, =, or > symbol to separate substances in the list.) (a) the Period 4 elements V, Ge, and K (b) the Group 5A elements N, As, and Bi Arrange in order of increasing atomic size. (Use the appropriate <, =, or > symbol to separate substances in the list.) (a) the Period 3 elements Mg, Si, and Ar (b) the Group 2A elements Ca, Ba, and Srarrow_forwardHow many inner, outer, and valence electrons are present in an atom of each of the following elements?(a) O(b) Sn(c) Ca(d) Fe(e) Searrow_forwardBoron, atomic number 5, occurs naturally as two isotopes, 10B and 11B, with natural abundances of 19.9% and 80.1%, respectively.(a) In what ways do the two isotopes differ from each other? Does the electronic configuration of 10B differ from that of 11B? (b) Drawthe orbital diagram for an atom of 11B. Which electrons are the valence electrons? (c) Indicate three ways in which the 1s electrons inboron differ from its 2s electrons. (d) Elemental boron reacts with fluorine to form BF3, a gas. Write a balanced chemical equation forthe reaction of solid boron with fluorine gas. (e) ΔHf° for BF31g2 is -1135.6 kJ>mol. Calculate the standard enthalpy change in thereaction of boron with fluorine. (f) Will the mass percentage of F be the same in 10BF3 and 11BF3? If not, why is that the case?arrow_forward
- Which of the following statements about the hydrogen atom is FALSE. (A) The hydrogen atom is neutral. (B)The hydrogen atom has a uniform distribution of charge. (C)The hydrogen atom is not polar. (D)The hydrogen atom is spherically symmetric.arrow_forwardArrange in order of increasing nonmetallic character. (a) the period 4 elements Ga, Ge, Ti (b) the Group 5A elements P, Bi, and Narrow_forward(c) Silicon (Si) is the most common chemical element in today's semiconductor industry. It has an atomic number of 14 and belongs to the Group IV (4) of the periodic table with its most common isotope being Si-29. (i) (ii) (iii) Explain what an isotope is. How many protons and how many neutrons are in the nucleus of this Silicon isotope? What is the electron configuration of Si?arrow_forward
- Which of these elements is most likely to form ions with a2+charge?(a) Li (b) Ca (c) O (d) P (e) Clarrow_forwardWrite the chemical formulas for the following compounds:(a) Silver cyanide(b) Calcium hypochlorite(c) Potassium chromate(d) Gallium oxide(e) Potassium superoxide(f) Barium hydrogen carbonatearrow_forwardThe oxygen and nitrogen families have some obvious similarities and differences.(a) State two general physical similarities between Group 5A(15) and 6A(16) elements.(b) State two general chemical similarities between Group 5A(15) and 6A(16) elements.(c) State two chemical similarities between P and S.(d) State two physical similarities between N and O.(e) State two chemical differences between N and O.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
