
(a)
Interpretation:
The atom has higher
The given diagram is,
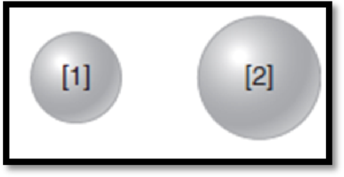
Figure 1
Concept Introduction:
Atomic number (Z):
Atomic number of an element is the number of protons found in the nucleus of every atom of that element.
(b)
Interpretation:
The atom has higher ionization energy has to be given.
The given diagram is,
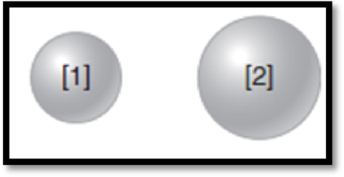
Figure 1
Concept Introduction:
Ionization energy:
Ionization energy is defined as the energy required removing one mole of electron from one mole of isolated gaseous atom or ion.
Periodic trends:
Ionization energy of an element increases along the period and decreases down the group.
(c)
Interpretation:
The atom has higher ionization energy has to be given.
The given diagram is,
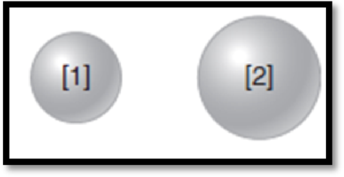
Figure 1
Concept Introduction:
Refer to part (b).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- 2.42 What is a period in the periodic table? From what does it derive its name?arrow_forwardRubidium and bromine atoms are depicted at right. (a) What monatomic ions do they form? (b) What electronic feature characterizes this pair of ions, and which noble gas are they related to? (c) Which pair best represents the relative ionic sizes?arrow_forwardQuestion:Consider two elements, Element A and Element B, from the periodic table. Element A is located in Group 1 and Period 3, while Element B is located in Group 16 and Period 2. (a) Which element would have a larger atomic radius, Element A or Element B? Justify your answer. (b) Predict the ionization energy of Element A and Element B. Which element would have a higher ionization energy? Explain your reasoning. (c) Based on their positions in the periodic table, which element would be more likely to form a stable cation? Provide a brief explanation for your choice.arrow_forward
- 2. (a) What do you understand by the statement: "the energy of the electrons in an atom is quantized"? (b) Why did Rutherford select alpha particles in his gold foil experiment? Briefly explain your answer in own words.arrow_forward(a) Write out the full electron configuration of Calcium (Ca). (b) How many electrons are in the n=3 shell of Calcium?arrow_forwardConsider the following information about three atoms: (a) How are they similar to one another? (b) How do they differ from one another? (c) What are the members of such a group of atoms called? (d) Using the planetary model, draw the atomic configuration of 126C showing the relative position and numbers of its subatomic particles.arrow_forward
- Which statement about electrons is false?(a) Most atoms have more electrons than protons.(b) Electrons have a charge of 1-.(c) If an atom has an equal number of protons and electrons,it will be charge-neutral.(d) Electrons experience an attraction to protons.arrow_forwardWhich of the following statements about the hydrogen atom is FALSE. (A) The hydrogen atom is neutral. (B)The hydrogen atom has a uniform distribution of charge. (C)The hydrogen atom is not polar. (D)The hydrogen atom is spherically symmetric.arrow_forwardArrange in order of increasing atomic size. (a) the period 3 elements Cl, Na, and Ar (b) the Group 2A elements Ca, Be, and Mgarrow_forward
- The graph at the right shows the atomic radius for the19 elements in the periodic table.(a) Describe the trend in atomic radius in going fromH (atomic number 1) to K (atomic number 19).(b) Find the three elements represented with blue dotson a periodic table. What do their placements in thetable have in common?(c) Find the three elements represented with red dotson a periodic table. What do their placements in thetable have in common?(d) Based on the graph, what is the radius of C?arrow_forwardExamine the ratio of atomic mass to atomic number for the elements with even atomic number through calcium. This ratio is approximately the ratio of the average mass number to the atomic number. (a) Which two elements stand out as different in this set of ten? (b) What would be the "expected" atomic mass of argon, based on the correlation considered here? (c) Show how the anomaly in the ordering of natural atomic masses of argon and potassium can be accounted for by the formation of “extra" 40 Ar via decay of 4°K atoms.arrow_forwardAn element has nine protons and 10 neutrons in the neutral atom. (a) How many electrons are present in the neutral atom? (b) What is the atomic number of this element? (c) Identify the element.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning  Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





