
Concept explainers
(a)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Bond order is calculated by the expression given as follows:
The magnetic properties are related to terms such as diamagnetism and paramagnetism. Paramagnetism defines the ability of elements to be weakly attracted in an external magnetic field. It arises due to the presence of unpaired electrons. Diamagnetism defines the ability to be repelled in the external magnetic field environment. This is because diamagnetic species have paired electrons.
The bond length is estimated to be an average of covalent radii of two atoms within a bond. When bond order increases bond becomes stronger and bond length reduces. This accounts for shorter bond length in the case of unsaturated compounds while longer bonds in saturated compounds.
(a)
Explanation of Solution
For
The corresponding molecular orbitals in
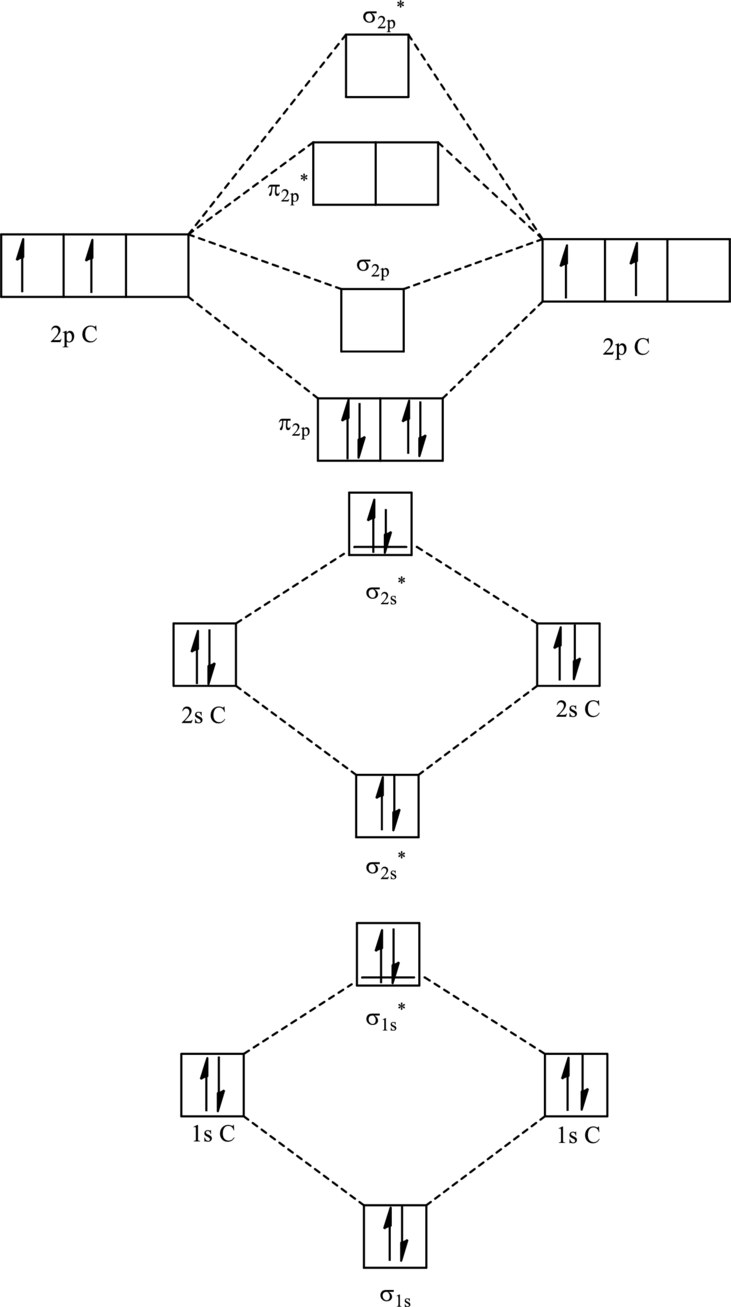
Bond order is calculated by the expression given as follows:
Substitute 4 for anti-bonding electrons and 8 for bonding electrons in equation (1).
Thus bond order is 2 in
For
Substitute 4 for anti-bonding electrons and 7 for bonding electrons in equation (1).
Thus bond order is 1.5 in
Since the reduction in bond implies bond is longer thus the bond length is more in case of
(b)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
For
The corresponding molecular orbitals in
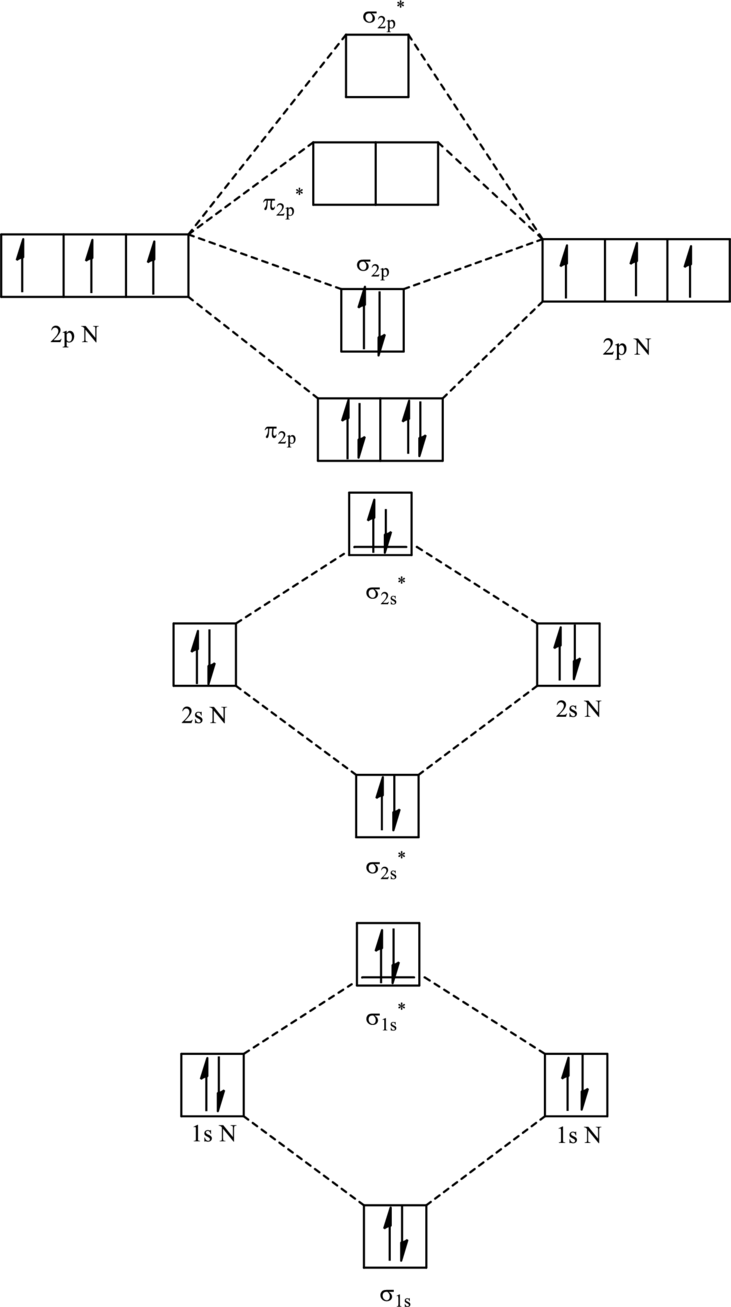
Bond order is calculated by the expression given as follows:
Substitute 4 for anti-bonding electron and 10 for bonding electrons in equation (1).
Thus bond order is 3 in
For
Substitute 4 for anti-bonding electrons and 9 for bonding electrons in equation (1).
Thus bond order is 2.5 in
Since the reduction in bond order implies bond is longer thus the bond length is more in case of
Further in
(b)
Interpretation:
Changes in bond order, bond distance, and magnetic properties expected when
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
For
The corresponding molecular orbitals in
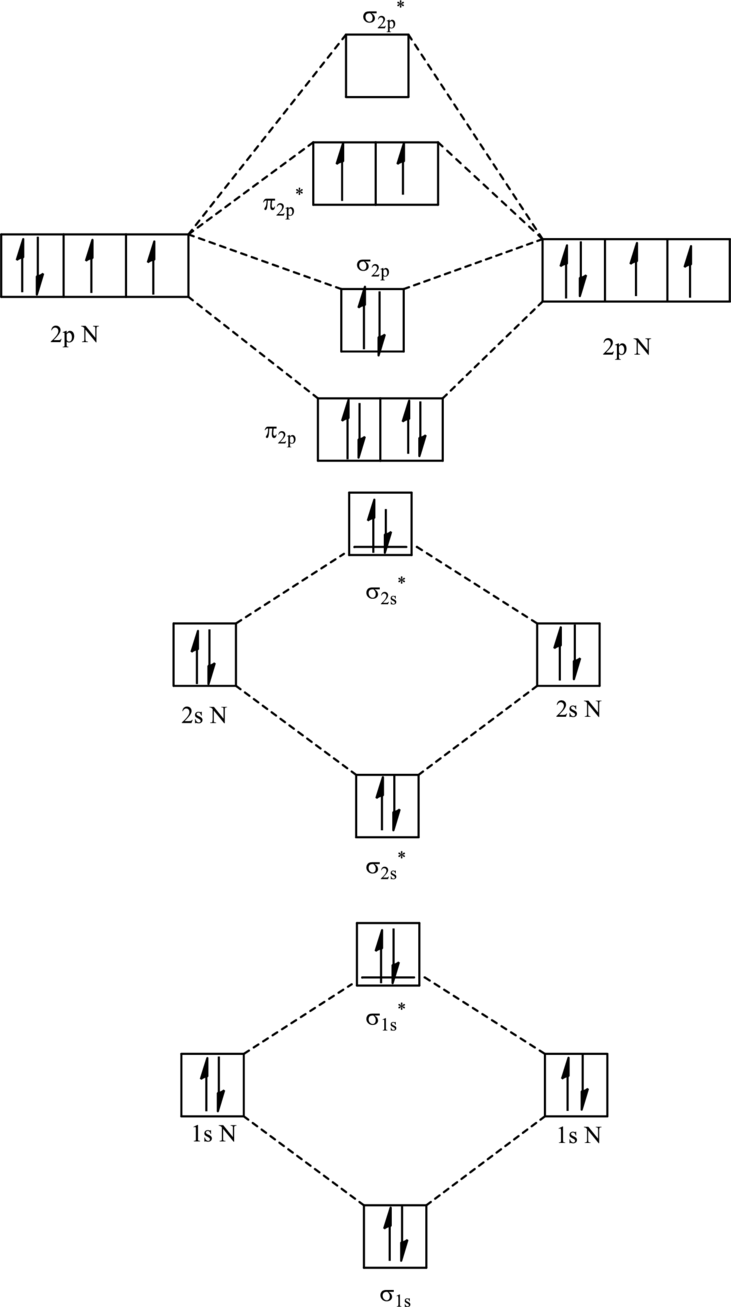
Bond order is calculated by the expression given as follows:
Substitute 6 for anti-bonding electrons and 10 for bonding electrons in equation (1).
Thus bond order is 2 in
For
Substitute 5 for anti-bonding electrons and 10 for bonding electrons in equation (1).
Thus bond order is 2.5 in
Since the reduction in bond order implies bond is shorter thus the bond length is more in case of
Further in
Want to see more full solutions like this?
Chapter 2 Solutions
CHEMICAL PRINCIPLES W/SAPLING
- The cations O2+ and N2+ are formed when molecules of O2 and N2 are subjected to intense, high-energy solar radiation in Earths upper atmosphere. Write the electron configuration for O2+. Predict its bond order and magnetic behavior.arrow_forwardFor the carbonate ion, CO3 2−, draw all of the resonance structures. Identify which orbitals overlap to create each bond.arrow_forwardWhat is the hybridization of the central atom in the sulfur trifluoride SF−3 anion?arrow_forward
- The bond between carbon and hydrogen is one of the most important typesof bonds in chemistry. The length of an H¬C bond is approximately 1.1 Å.Based on this distance and differences in electronegativity, do you expect thedipole moment of an individual H¬C bond to be larger or smaller than that ofan H¬I bond?arrow_forwardHow many double bonds can be found in the best Lewis stucture of COF 2?arrow_forwardElectrons in bonds are negatively charged and therefore repel each other. If you had a molecule of formula AB 2 , with A being the central atom, what would you predict the 3D shape to be to allow maximum distance between the two B atoms bonded to A?arrow_forward
- Account for the ability of phosphorus to form five bonds, as in PF5.arrow_forwardDescribe the electron-pair geomtery and molecular geomtery around the central atom in GeBr3-arrow_forwardIf an element is bonded to 4 other atoms and has a formal charge of +1, what group must the element be in? I know that group 3A atoms are elctron deficient, and that period 3 elements and below, except for group 3A elements like Aluminum, can expand their octet because of their available d-orbital, which may not be relevant to this problem. I don't understand this question, or why the answer would be 5A. Is it because 5A have odd valence electrons, and can form free radicals, like NO?arrow_forward
- What are the relative lengths and strengths of the bonds in Cl2 and Br2?arrow_forwardWhich is the most likely molecular structure for the nitrite (NO2-) ion?arrow_forwardDraw the Lewis structure of HClO₃ (with minimized formal charges) and then choose the appropriate pair of molecular geometries of the two central atoms. Your answer choice is independent of the orientation of your drawn structure.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


