
Interpretation:
The group, period, metallic character, number of protons, name of the element and the electron dot symbol from the given electron configuration of an element should be identified.
Concept introduction:
The periodic table is shown in figure 1,
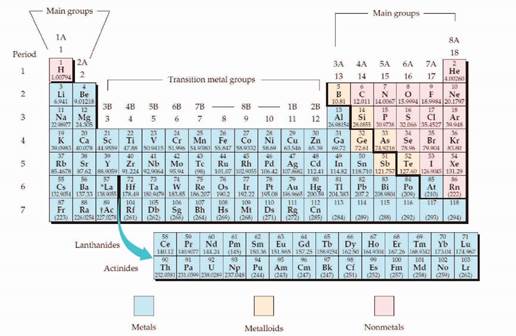
Figure 1
The Aufbau principle:
The aufbau principle is states that in the ground state of an atom, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied.
The subshell ordering by this rule is
Number of electrons in the shells is given below,
Each shell contains only a fixed number of electrons. The first shell contains two electrons, the second shell contains eight
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- How many oxygen atoms are in this molecular formula? 3H2 Oarrow_forwardExplain why lung and soft tissue appear different on a radiograph when they have both an effective atomic number of 7.4?arrow_forwardChlorine is an element with the symbol Cl. Draw and label atomic structure of chlorine indicating the number and locations of protons, neutrons and electrons in one atom of chlorine.arrow_forward
- Look up antimony in the periodic table 1Z = 512. How many covalent bonds would you expect it to form? Based on this information, which of the following antimony compounds is covalent and which is ionic: SbCl3 or SbCl5?arrow_forwardIf you are told you have an element with an atomic number of 16 and its in group VIA. What does this tell you about the number of protons, neutrons and electrons? What does it tell you about its valance electrons and will it tend to form anions or cations? Is it a metal or a nonmetal?arrow_forwardWhich electron shell is the valence shell of an atom, and what is its significance?arrow_forward
- Complete the table below, using the diagram of an atom shown at right. name symbol 0 proton e Properties of subatomic particles charge (in multiples of e) 0 0 0 approximate mass (amu) (choose one) ✓ (choose one) ✓ 1.0 location on diagram (choose one) ✓ (choose one) ✓ A X A Sarrow_forwardHow many electrons are in the outer shell of each of the following atoms?arrow_forwardWhich statements are true regarding the chemical formula Pb(NO3)4. Select the three that apply. The formula unit has a ratio of four nitrate ion for every lead (IV) ion. Each Nitrogen atom is bonded with three oxygen atoms Formula unit contains more lead (IV) ions than nitrate ions. Each nitrate ion is bonded with four lead (IV) ions. Each formula unit has total of seventeen atomsarrow_forward
- Why do the 3s, 3p, and 3d orbitals have the same energy in a hydrogen atom but different energies in a many- electron atom?arrow_forwardWhat is the mass in grams of 6.022 * 1023 O atoms of mass 16.00 amu?arrow_forwardPolonium is a rare element with 33 radioisotopes. The most common one, 210Po, has 82 protons and 128 neutrons. When 210Po decays, it emits an alpha particle, which is a helium nucleus (2 protons and 2 neutrons). 210Po decay is tricky to detect because alpha particles do not carry very much energy compared to other forms of radiation. They can be stopped by, for example, a sheet of paper or a few inches of air. This property is one reason why authorities failed to discover toxic amounts of 210Po in the body of former KGB agent Alexander Litvinenko until after he died suddenly and mysteriously in 2006. What element does an atom of 210Po decay into after it emits an alpha particle?arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning





