
Concept explainers
(a)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
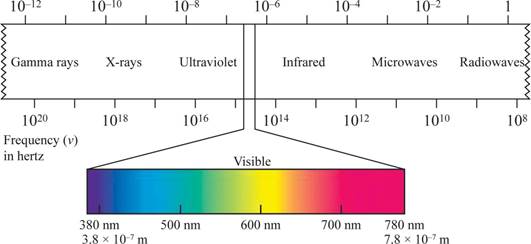
Figure 1
(b)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
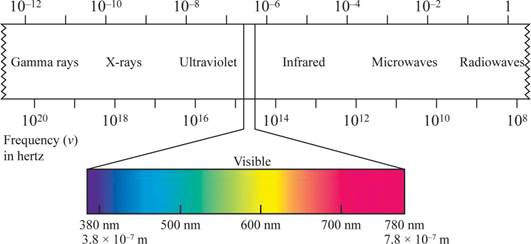
Figure 1
(c)
Interpretation:
The higher energy should be identified.
Concept introduction:
The shorter the wavelength, the higher the energy, the longer the wavelength, the lower the energy.
Wave length region is shown in figure 1.
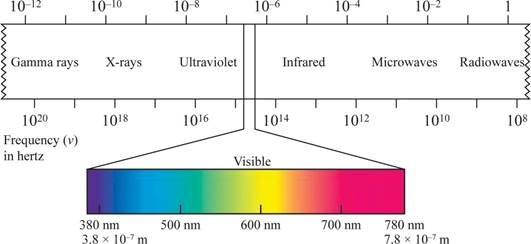
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- (a) How much time is needed to measure the kinetic energy of an electron whose speed is 10.0 m/s with an uncertainty of no more than 0.100 percent? How far will the electron have traveled in this period of time? (b) Make the same calculations for a 1.00-g insect whose speed is the same. What do these sets of figures indicate?arrow_forwardThe magnesium electron spectrum has a line at 266.8 nm. Does it have a greater speed in a vacuum (an area without an atom or particle) than does red light with a wavelength of 652 nm? Why or why not?arrow_forwardAt the beginning of an experiment, a scientist has 268 grams of radioactive goo. After 180 minutes, her sample has decayed to 4.1875 grams.What is the half-life of the goo in minutes? Find a formula for G(t)G(t), the amount of goo remaining at time tt. How many grams of goo will remain after 32 minutes?arrow_forward
- A slice of pizza has 500 kcal. If we could burn the pizza and useall the heat to warm a 50-L container of cold water, what wouldbe the approximate increase in the temperature of the water?(Note: A liter of cold water weighs about 1 kg.)(A) 50°C (B) 5°C(C) 100°C (D) 10°Carrow_forwardIf a hospital were storing radioisotopes, what is the minimum containment needed to protect against:(a) cobalt-60 (a strong γ emitter used for irradiation)(b) molybdenum-99 (a beta emitter used to produce technetium-99 for imaging)arrow_forwardWhich of the following types of photons (packets of pure electromagnetic energy) have the highest energy level? visible light infrared microwaves ultra-violetarrow_forward
- In your own words define what is Dark matter?arrow_forwardIf there is 10 μmol of the radioactive isotope 32P (half-life 14 days) at t = 0, how much 32P will remain at (a) 7 days, (b) 14 days, (c) 21 days, and (d) 70 days?arrow_forward(I) Why the physicochemical changes in liquid water caused by radiation is the key to understanding the biological effects of radiation? Please give a short, one-sentence explanation. (II) Name the three key stages of the physicochemical changes produced in liquid water due to radiation. (III) Briefly describe the three key stages. A one- or two-sentence explanation for each of the three key stages would be sufficient.arrow_forward
- If a particle in an electric field E experiences a force F, what also must be true?A)The particle has a nonzero charge.B) The particle is positively charged.C) The particle is negatively charged.D) The particle is moving.arrow_forwardThis is about Beer-Lambert’s Law and UV-vis Spectroscopy: 1) How does a solution interact with light?2) How are absorbance and transmittance related? 3) How do you make an absorbance spectrum?arrow_forwardThe difference in chemical potential of a particular substance between two regions of a system is −8.3 kJ mol−1. By how much does the Gibbs energy change when 0.15 mmol of that substance is transferred from one region to the other?arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
