
(a)
Interpretation:
The group, period and the metallic character of element should be identified.
Concept introduction:
The periodic table is given below,
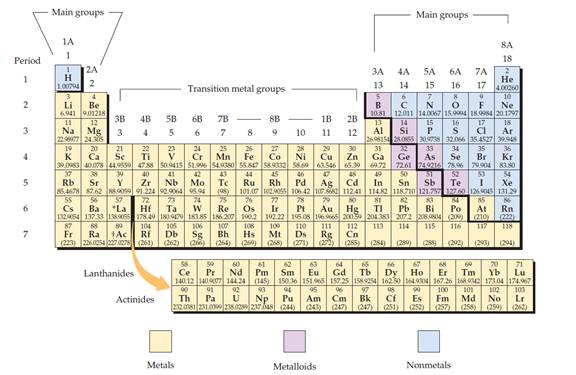
Figure 1
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals.
Metal:
A metal is a material that, when freshly prepared, polished, or fractured, has a shiny appearance. Metal has the capacity to conducts the electricity and heat. Metals are malleable or ductile.
Non-metal:
Non-metal is an element that mostly absences metallic character.
Metalloid:
Metalloid is one of the types of chemical element, which has properties in between, or that are a mixture of metals and nonmetals.
Alkali metals:
Alkali metals are a group (column) in the periodic table is lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr).
Alkaline earth metals:
Alkaline earths metals are group two of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).
(b)
Interpretation:
The group, period and the metallic character of element should be identified.
Concept introduction:
The periodic table is given below,
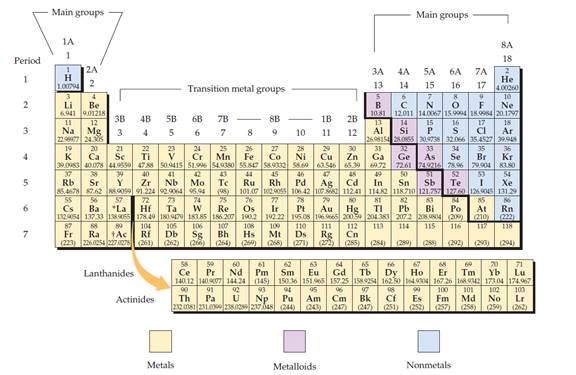
The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals.
Metal:
A metal is a material that, when freshly prepared, polished, or fractured, has a shiny appearance. Metal has the capacity to conducts the electricity and heat. Metals are malleable or ductile.
Non-metal:
Non-metal is an element that mostly absences metallic character.
Metalloid:
Metalloid is one of the types of chemical element, which has properties in between, or that are a mixture of metals and nonmetals.
Alkali metals:
Alkali metals are a group (column) in the periodic table is lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr).
Alkaline earth metals:
Alkaline earths metals are group two of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- Look up the valence electron configuration, covalent atomic radius, effective nuclear charge, first ionization energy and Pauling electronegativity in Chapter 8 (tables are attached). Examine the above data and answer the following questions. a) Explain why some of the elements like TI and Pb on the lower left of the p block are metallic. b) Explain why some of the elements like C, Si in the center of the p block form covalent bonds. Explain why these bonds formed by the network of these elements (as studied in Chapter 25) tend to be unreactive. c) Explain why the noble Group 8A elements are highly unreactive gases. d) Explain why some elements like F, CI, Br etc, on the upper right of the p block are highly reactive nonmetals.arrow_forwardA lithium atom has 3 protons and 4 neutrons. What is itsmass number?arrow_forwardA compound with empirical formula C2H5O was found in a separate experiment to have a molar mass of approximately 90 g. What is the molecular formula of the compound?arrow_forward
- Explain why lung and soft tissue appear different on a radiograph when they have both an effective atomic number of 7.4?arrow_forwardWhat is the mass in grams of 6.022 * 1023 O atoms of mass 16.00 amu?arrow_forwardElements have varying numbers of protons, neutrons, and electrons.True or false?arrow_forward
- Compound A has molecular formula C7H7X. Its 1H-NMR spectrum shows a singlet at 2.25 ppm and two doublets, one at 7.28 ppm and one at 7.39 ppm. The singlet has an integral of three and the doublets each have an integral of two. The mass spectrum of A shows a peak at m/z = 126 and another peak at m/z = 128; the relative height of the two peaks is 3:1 respectively. Identify what atom X is, explaining your reasoning and identify Compound A, explaining your reasoning.arrow_forwardHow does the atomic structure or composition of the versions of sodium in question C above differ from a typical sodium atom, with its atomic mass of 23?arrow_forwardLook up antimony in the periodic table 1Z = 512. How many covalent bonds would you expect it to form? Based on this information, which of the following antimony compounds is covalent and which is ionic: SbCl3 or SbCl5?arrow_forward
- Arrange in order of increasing atomic size. (a) the period 2 elements Li, C, and F (b) the group 4A elements Sn, Ge, and Siarrow_forwarda) Identify the following as element (atomic or molecular), compound or mixture. b) Explain your reasoning and draw a sample of these substances containing 5 particles of that particular material. c) What would be the mass of 5 particles of that particular material? Show your work! H20 N2 C2HSOH Iodine gasarrow_forwardIdentify the names of the following structure. (with alpha/beta and L-D designation)arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

