
Concept explainers
(a)
Interpretation:
The VSEPR formula and shape for sulfur tetrachloride molecule have to be predicted.
Concept Introduction:
Valence Shell Electron Pair Repulsion model predicts shape by inclusion of bond angles and most distant arrangement of atoms that leads to minimum repulsion. For the molecules that have no lone pairs around the central atom the bonded-atom unshared -pair arrangement is decided by the table as follows:
In order to determine the shape the steps to be followed are indicated as follows:
- 1. Lewis structure of molecule should be written.
- 2. The type electron arrangement around the central atom should be identified around the central atom. This essentially refers to determination of bond pairs and unshared or lone pairs around central atoms.
- 3. Then bonded-atom unshared -pair arrangement that can maximize the distance of electron pairs about central atom determines the shape.
For molecules that have lone pairs around central atom, lone pairs influence shape, because there are no atoms at the positions occupied by these lone pairs. The key rule that governs the molecular shape, in this case, is the extent of lone –lone pair repulsions are far greater than lone bond pair or bond pair-bond pair repulsions. The table that summarized the molecular shapes possible for various combinations of bonded and lone pairs are given as follows:
(a)
Answer to Problem 2E.11E
The shape for sulfur tetrachloride molecule is square planar and VSEPR formula is
Explanation of Solution
Sulfur tetrachloride has sulphur as central atom. Sulfur has six valence electrons while chlorine possesses seven valence electrons.
Total valence electrons are sum of the valence electrons on each chlorine and central sulfur in
The skeleton structure in
These 13 electron pairs are assigned as lone pairs of each of the chlorine atoms to satisfy its octet.
Hence, the Lewis structure of
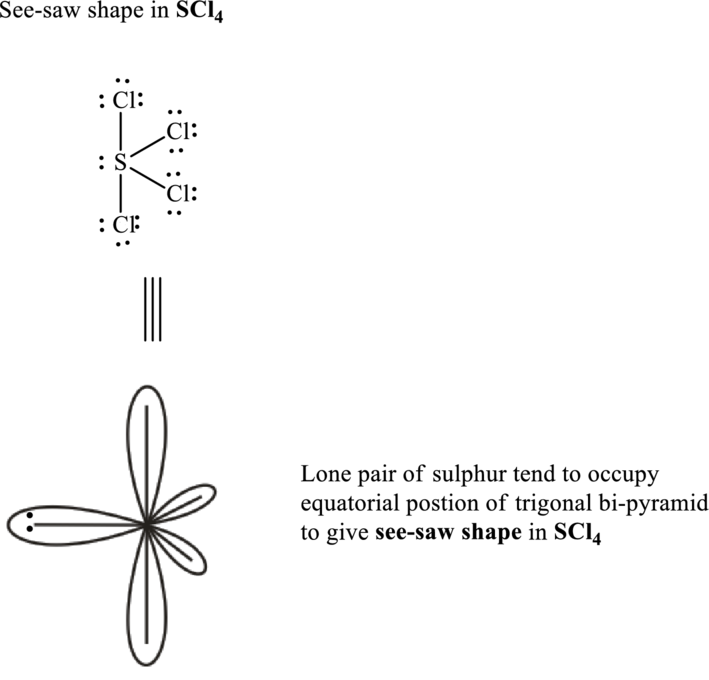
It is evident that in
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any square planar species the VSEPR formula is predicted as
(b)
Interpretation:
The VSEPR formula and shape for iodine trichloride molecule have to be predicted.
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 2E.11E
The shape for iodine trichloride is T-shape and VSEPR formula is
Explanation of Solution
Iodine trichloride has
Total valence electrons are sum of the valence electrons on each chlorine and central iodine in
The skeleton structure in
These 11 electron pairs are allotted as lone pairs of each of the chlorine atoms to satisfy its octet. Hence, the Lewis structure and corresponding T-shape in
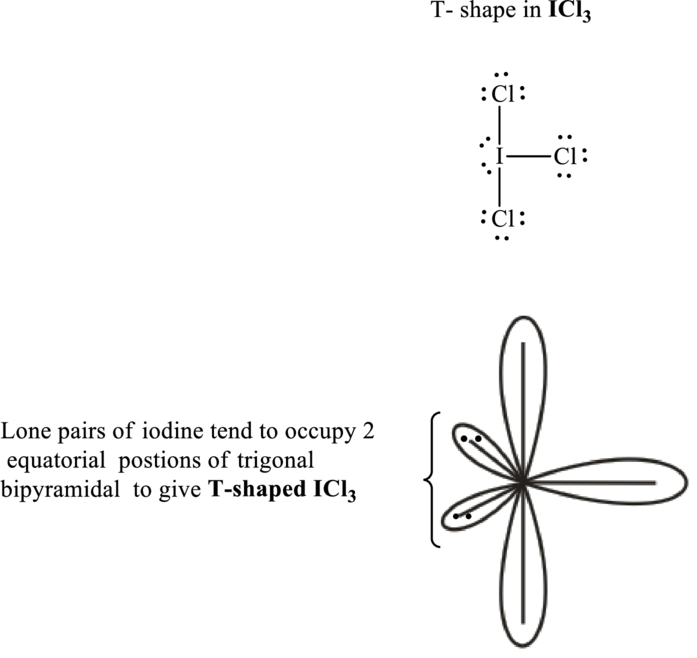
It is evident that in
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any bent or T-shaped species the VSEPR formula is predicted to be
(c)
Interpretation:
The VSEPR formula and shape for
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 2E.11E
The shape for
Explanation of Solution
Total valence electrons are sum of the valence electrons on each fluorine and central iodine in
The skeleton structure in
These 14 electron pairs are allotted as lone pairs of each of the fluorine atoms to satisfy its octet. Hence, the Lewis structure and corresponding T-shape in
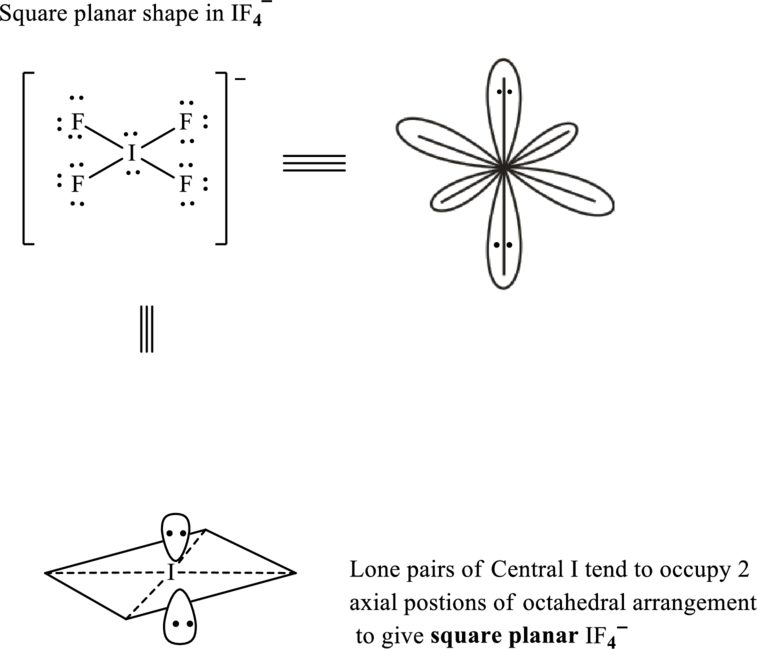
In
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for any square planar species the VSEPR formula is predicted as
(d)
Interpretation:
The VSEPR formula and shape for xenon trioxide molecule have to be predicted.
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 2E.11E
The shape for xenon trioxide molecule is trigonal pyramidal and corresponding VSEPR formula is
Explanation of Solution
Xenon trioxide has
Total valence electrons are sum of the valence electrons on each oxygen atom and central
The skeleton structure in
These 7 electron pairs are allotted as lone pairs of each of the oxygen atoms to satisfy its octet. Thus, the Lewis structure and shape of
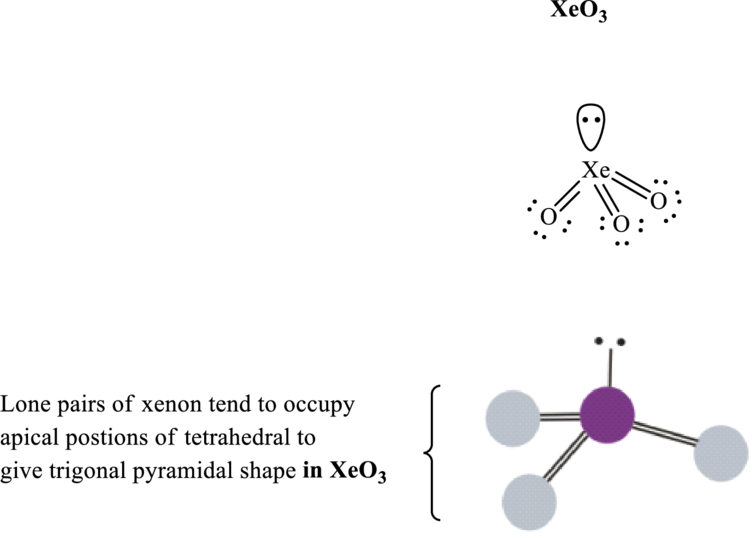
It is evident that in
If lone pairs are represented by E, central atom with A and other attached bond pairs by X, then for trigonal pyramidal any species the VSEPR formula is predicted as
Want to see more full solutions like this?
Chapter 2 Solutions
CHEMICAL PRINCIPLES 2017 PKG
- Write the Lewis structures for the following species, and indicate whether each is an odd-electron species, an electron-deficient species, or an expanded valence shell species. (a) BI3 (b) IF5 (c) HN2arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forwardHow many double bonds can be found in the best Lewis stucture of COF 2?arrow_forward
- What are the favored geometrical arrangements for ABn molecules for which the A atom has 2, 3, 4, 5, and 6 pairs of electrons in its valence shell?arrow_forwardDichlorine heptaoxide, Cl₂O₇, can be viewed as two ClO₄ groups sharing an O atom. Draw a Lewis structure for Cl₂O₇ with the lowest formal charges, and predict any deviation fromthe ideal for the Cl-O-Cl bond angle.arrow_forwardDraw the Lewis structure of the azide ion, N3, and calculate the formal charge on each nitrogen atom in the structure:arrow_forward
- Draw resonance structures for the formate ion, HCO2- and then determine the C-O bond order in the ion.arrow_forwardWhat is the electron-pair geometry around the central S atom in sulfuryl fluoride, SO2F2?arrow_forwardChemical species are said to be isoelectronic if they have the same Lewis structure (regardless of charge). Consider these ions and write a Lewis structure for a neutral molecule that is isoelectronic with them. (a) CN–, (b) NH4+ (c) CO3 2–arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


