
Concept explainers
(a)
Interpretation:
Hybridization of underlined
Concept Introduction:
Hybridization is the hypothetical concept of mixing of atomic orbital into hybrid orbitals that are of dissimilar shapes, energies and are appropriate for combination of electrons to form bonds in
Hybridization is calculated by the hybrid orbitals and to calculate hybrid orbitals we need to know the steric number that is given by,
The table that relates the steric number with hybridization is as follows:
(a)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
Its Lewis structure is as follows:
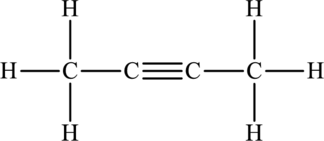
As from the structure above, 2 atoms are bonded to the underlined atom and 0 lone pair on it.
Substitute 2 for number of atoms bonded to the central atom and 0 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(b)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
The symbol for nitrogen is
Its Lewis structure is as follows:
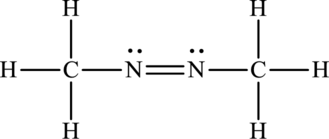
As from the structure above, 2 atoms are bonded to the underlined nitrogen atom and 1 lone pair on it.
Substitute 2 for a number of atoms bonded to the central atom and 1 for a number of lone pairs on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(c)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
Its Lewis structure is as follows:
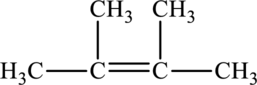
As from the structure above, 3 atoms are bonded to the underlined carbon atom and 0 lone pair on the same atom.
Substitute 3 for a number of atoms bonded to the central atom and 0 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
(d)
Interpretation:
Hybridization of underlined
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 2F.8E
The molecule
Explanation of Solution
The molecule
The symbol for carbon is
The symbol for hydrogen is
The symbol for nitrogen is
Its Lewis structure is as follows:
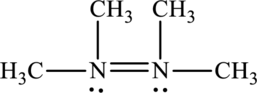
As from the structure above, 3 atoms are bonded to the underlined nitrogen atom and 1 lone pair on the same atom.
Substitute 3 for a number of atoms bonded to central atom and 1 for a number of lone pair on the central atom in equation (1) to find out the steric number.
Hence, the hybridization of
Want to see more full solutions like this?
Chapter 2 Solutions
CHEMICAL PRINCIPLES 2017 PKG
- trans-tetrazene (N4H4) consists of a chain of four nitrogen atoms with each of the two end atoms bonded to two hydrogen atoms. Use the concepts of steric number and hybridization to predict the overall geometry of the molecule. Give the expected structure of cis-tetrazene.arrow_forwardAspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forwardWhy do we hybtidize atomic orbitals to explain the bonding in covalent compounds? What type of bonds form from hybrid orbitals: sigma or pi? Explain.arrow_forward
- Give the hybridization of each central atom in the following molecules. (a) cyclohexene (b) phosgene, Cl2CO (c) glycine, H2NC(1)H2C(2)OOH (Note: Numbers in parentheses label each carbon atom.)arrow_forwardLabel and sketch the shape of hybrid orbitals of the central atom of PCI5arrow_forwardPlease give the hybridization, arrangement, and number of sigma and pi bonds in C2H3Cl and C5H6. Note any differences in the hybridization.arrow_forward
- a) Construct the molecular orbitals of NO-. b) Between the molecular HNO and HON, on the basis of your estimation and your answer to part a, which structure is favored?arrow_forwardRank the following molecules by order of increasing bond energy: F2, F2^+, F2^2+. Justify your answer in detail.arrow_forwardDemonstrate the hybridization types of carbon, oxygen and nitrogen atoms in the following molecule and show their 3D states by considering their bond angles. H3C-CH=CH-C≡CHarrow_forward
- How many hybrid orbitals are represented in the picture, hoe many s,p,d atomic orbitals corresponding to what hybridization, what electron pair, and what bond angle(s)?arrow_forwardComplete the following table by providing theVSEPR-name and hybridisation (sp, sp2 or sp3) ofthe atom indicated by the arrow.arrow_forwardAllene (1,2-propadiene), H2C=C=CH2, has two adjacent double bonds. What kind of hybridization must the central carbon have? Sketch the bonding π orbitals in allene.What shape do you predict for allene?arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





