
EBK BIOLOGY TODAY AND TOMORROW WITHOUT
5th Edition
ISBN: 8220100557187
Author: STARR
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 2FIO
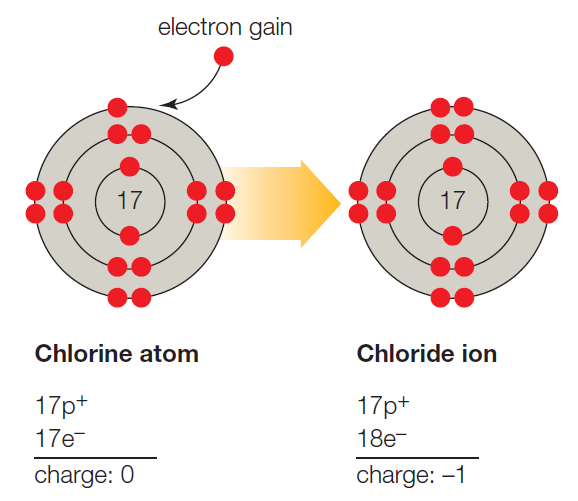
B. A chlorine atom (Cl) becomes a negatively charged chloride ion (Cl−) when it gains an electron and fills the vacancy in its third, outermost shell.
Figure It Out: Does a chloride ion have an unpaired electron?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Page 16: Draw a picture of a pH scale. Be sure to label the following terms or
your picture: acid, base, neutral, high concentration of hydrogen (H*), & low
concentration of hydrogen (H+).
A. Write the products of each of the following reactions and balance the equation. Indicate
for cach product whether it is solid (s), liquid (), aqueous (aq) or gas (g). If no reaction
occurs, the products are the same as the reactants, write no reaction and proceed to the
next reaction.
B. Write an ionic equation for each reaction. Break up only compounds with (aq) into ions.
Recall that the number of ions in a compound are indicated with a subscript, but the
number of ions listed separately in an equation are indicated with a coefficient.
C. Cancel out any ions that appear on both sides of the equation and write the net ionic
equation for each.
Describe ionization and distinguish between anions and cations.
Chapter 2 Solutions
EBK BIOLOGY TODAY AND TOMORROW WITHOUT
Ch. 2 - A. The first shell corresponds to the first energy...Ch. 2 - B. A chlorine atom (Cl) becomes a negatively...Ch. 2 - Figure 2.12 A pH scale. Here, red dots signify...Ch. 2 - Figure 2.17 Fatty acids. Double bonds in the tails...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Prob. 1SQCh. 2 - Which element has only one proton?Ch. 2 - The mutual attraction of opposite charges holds...
Ch. 2 - A salt does not release __________ in water. a....Ch. 2 - A(n) _______ substance repels water. a. acidic b....Ch. 2 - When dissolved in water, a(n) _____ donates H+ and...Ch. 2 - _________ is a monosaccharide. a. Glucose b....Ch. 2 - Unlike saturated fatty acids, the tails of...Ch. 2 - Which of the following is a class of molecules...Ch. 2 - Prob. 10SQCh. 2 - Prob. 11SQCh. 2 - Prob. 12SQCh. 2 - Prob. 13SQCh. 2 - Match the molecules with the best description.Ch. 2 - Match each molecule with its component(s).Ch. 2 - Alchemists were the forerunners of modern-day...Ch. 2 - Prob. 2CTCh. 2 - Polonium is a rare element with 33 radioisotopes....Ch. 2 - In the following list, identify the carbohydrate,...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- . Complete the following diagram illustrating ionic bonds. Atom X gives one electron to each atom Y. What are the ions that form as a result? Put the charges under each atom. Atom Atom Atorn Y Yarrow_forwardA solution at pH 7 is considered neutral because (a) its hydrogen ion concentration is 0 mol/L (b) its hydroxide ion concentration is 0 mol/L (c) the product of its hydrogen ion concentration and its hydroxide ion concentration is 0 mol/L (d) its hydrogen ion concentration is equal to its hydroxide ion concentration (e) it is nonpolararrow_forwardAnother fossil-fuel gas used in cooking appliances is "propane", used commonly in backyard grills and RV/camping stoves. Propane is a bigger hydrocarbon: 3 carbons single-bonded in a chain, and the remaining bonds are taken by hydrogen. Its formula is C3H8 and its structure is shown below. KEY POINT: the products are the same as the combustion of methane, but more is produced. The graphic below shows that one propane molecule reacts with 5 oxygen molecules to form molecules of carbon dioxide. C3Hg + 502 → 3CO,+ 4H,0 00O 000 SUOLDICIATEFIEETILNED I RLIANMr CASIRICTIOonL Harrow_forward
- 40C. NAME this structure. Carrow_forwardExamine the chemical equation below, and label each reactant and product. (which is a reactant and product) H2O + CO2 ⇋ H2CO3 ⇋ HCO3- + H+ If you are blowing CO2 from your lung into a water solution, will it make the solution more acidic or basic?arrow_forwardExplain why oil does not dissolve in water ?arrow_forward
- нннн ннннН с-С-с-с-с-С-С-С-С-с-н HHHH HHHH H H. Ising the graphic above, answer the following question. orrectly identify the above molecule. s this molecule solid or liquid at room temperature? Name a major source of this macromolecule.arrow_forwardMention several essential ions found in living beings.arrow_forwardExplain the production of hydrogen bonds (give examples).arrow_forward
- if tomato juice has a pH of 3, how many more H+ ions are present in lemon juice with a pH of 4?arrow_forwardd.Which pairs are enantiomers? Which are diastereomers? e.Determine the absolute configuration of each chiral center in one pair of diastereomer.arrow_forwardAt pH=2.2, which of the following is true COOH Ç00 COO ÇO0 H3N-CH H,N-CH H,N-CH H2N-CH CH2 CH2 CH CH2 pK1 CH2 pKr CH2 pK, CH2 CH COOH СООН CO COO 1. II IV O50 % of carboxylic acid group in the side chain is ionized O Most of the molecules are in the form II C None 50% of carboxylic acid group attached to alpha carbon is ionized 50% of amino group is ionizedarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
What is a Primary and Secondary Metabolite?; Author: Unicity International;https://www.youtube.com/watch?v=TRNUURm0agM;License: Standard Youtube License