
EBK BIOLOGY TODAY AND TOMORROW WITHOUT
5th Edition
ISBN: 8220100557187
Author: STARR
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 3FIO
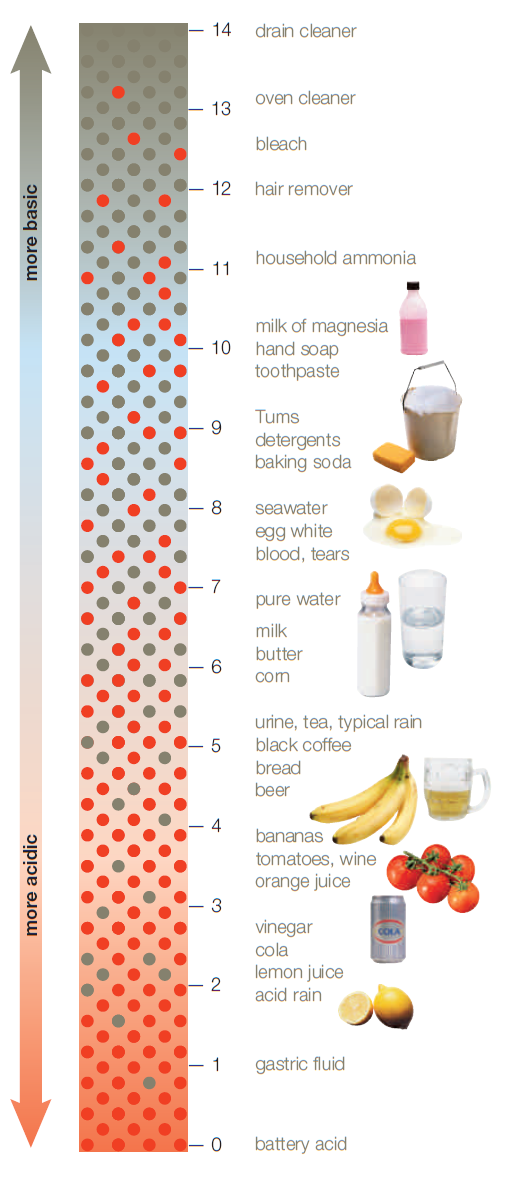
Figure 2.12 A pH scale.
Here, red dots signify hydrogen ions (H+) and blue dots signify hydroxyl ions (OH–). Also shown are the approximate pH values for some common solutions.
This pH scale ranges from 0 (most acidic) to 14 (most basic). A change of one unit on the scale corresponds to a tenfold change in the amount of H+ ions.
Photos, © JupiterImages Corporation.
Figure It Out: What is the approximate pH of cola?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
نقطة واحدة
The pH value is calculated
mathematically as the Log of the
* .hydrogen ion (H+) concentration
False
True O
an atom (to form cations) or adding electrons (to form anions). Ions are never formed by
1. It is important to recognize that ions are always formed by removing electrons from
a. There must be both positive ions (cations) and negative ions (anions) present.
2. It is very important to remember that a chemical compound must have a net charge
Read the following sentences.
changing the number of protons in an atom's nucleus.
of zero. This means that if a compound contains ions, then
a
Part I. For each of the following four questions: calculate and describe how the requested
solution would be made. Please show all of your calculations. In all cases the diluent or
solvent will be water. Also, assume the smallest volume you can accurately pipette is 1ul.
(CaClh: Molecular weight = 111.0 - NaCl: Molecular weight= 58.44)
1. From a 3M CaCh stock solution, you need to make 600 ml of 9 mM CaClh. Indicate how
you would make it.
2. You have a 20ul sample of DNA that you want to run in a gel. You are given 5X track
dye; how much track dye do you add to your sample so that the track dye concentration in
the sample is 1X?
3. You need to make 6 liters of 20% NaCl solution. Indicate how you wvould make it firom
powdered NACI.
Chapter 2 Solutions
EBK BIOLOGY TODAY AND TOMORROW WITHOUT
Ch. 2 - A. The first shell corresponds to the first energy...Ch. 2 - B. A chlorine atom (Cl) becomes a negatively...Ch. 2 - Figure 2.12 A pH scale. Here, red dots signify...Ch. 2 - Figure 2.17 Fatty acids. Double bonds in the tails...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Effects of Dietary Fats on Lipoprotein Levels...Ch. 2 - Prob. 1SQCh. 2 - Which element has only one proton?Ch. 2 - The mutual attraction of opposite charges holds...
Ch. 2 - A salt does not release __________ in water. a....Ch. 2 - A(n) _______ substance repels water. a. acidic b....Ch. 2 - When dissolved in water, a(n) _____ donates H+ and...Ch. 2 - _________ is a monosaccharide. a. Glucose b....Ch. 2 - Unlike saturated fatty acids, the tails of...Ch. 2 - Which of the following is a class of molecules...Ch. 2 - Prob. 10SQCh. 2 - Prob. 11SQCh. 2 - Prob. 12SQCh. 2 - Prob. 13SQCh. 2 - Match the molecules with the best description.Ch. 2 - Match each molecule with its component(s).Ch. 2 - Alchemists were the forerunners of modern-day...Ch. 2 - Prob. 2CTCh. 2 - Polonium is a rare element with 33 radioisotopes....Ch. 2 - In the following list, identify the carbohydrate,...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- From your data in Table 8.2 choose the correct pH for household chemical listed here. pH 6 purple pH 4 darker 1. Lemon juice pink 2. Mr. Clean pH 10- 12 3. Shampoo bluish green 4. Baking soda solution pH 10 teal or blue 5. Vinegar pH 2 lighter pinkarrow_forwardA solution with pH 4 has __________ the H+ concentration of pH 8.arrow_forwardThe pH scale shows availability of reactive hydrogen ions (H+) in a liquid. The scale is logarithmic, so milk has 10 times as many H+ ions as pure water, for a given volume. How many more H+ ions does soda pop have compared to pure water? Write the number only.arrow_forward
- The pH of a solution is 7, what is the H+ concentration? Group of answer choices 1 x 10e-7 M 1 x 10e-3 M 1 x 10e-8 M 1 x 10e-9 Marrow_forwardplease do not give solution in image format thanku Refer to the graph below, showing surface area and volume as functions of body size. Because of the relationships shown in the grapharrow_forwardTable 2. Volume of BSA, protein content, and absorbance readings of reference solutions Solution Volume of BSA standard solution (μL) Protein content(μg/mL) Absorbance value At 595 nm 1 0 0 0 2 10 1 0.022 3 30 3 0.065 4 50 5 0.106 5 70 7 0.178 6 100 10 0.299 7 120 12 0.380 Make a graph by plotting the absorbance values versus the BSA protein content (in μg) for theseven reference solutions. When constructing the graph, be…arrow_forward
- A solution in which ["OH] = 6.3 × 10-/ M has a higher pH than a solution with a ["OH] = 4.3 × 10-2 M. True Falsearrow_forwardA solution at pH 4 is _____ times as _______ as a solution at pH 7.arrow_forwardplese don't handwritten solution.. Compare the antacid properties of Magnesium Carbonate and indicate its advantages and disadvantages when compared to Magnesium Hydroxide Magnesium Trisilcate Magnesium Oxidearrow_forward
- Lemon juice has a pH around 2.5. Milk has a pH around 6.5-7. Which of the following statements about these two liquids is true? Lemon juice has a higher concentration of hydrogen ions compared to milk Milk has a lower concentration of hydroxyl ions compared to lemon juice Milk has a higher concentration of hydrogen ions compared to lemon juice The concentration of water in lemon juice and milk is identicalarrow_forwardShow solution. 3.arrow_forwardA solution with a pH of 6 has a ____ difference in H ion concentration than a solution with a pH of 10. If a solution has a concentration of 10^-7 OH ions, how many H ions does it have?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning

GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license