
Microbiology: An Introduction
12th Edition
ISBN: 9780321929150
Author: Gerard J. Tortora, Berdell R. Funke, Christine L. Case
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 3R
What type of bond holds the following atoms together?
- a. Li+ and Cl− in LiCl
- b. carbon and oxygen atoms in methanol
- c. oxygen atoms in O2
- d. a hydrogen atom of one
nucleotide to a nitrogen or oxygen atom of another nucleotide in:
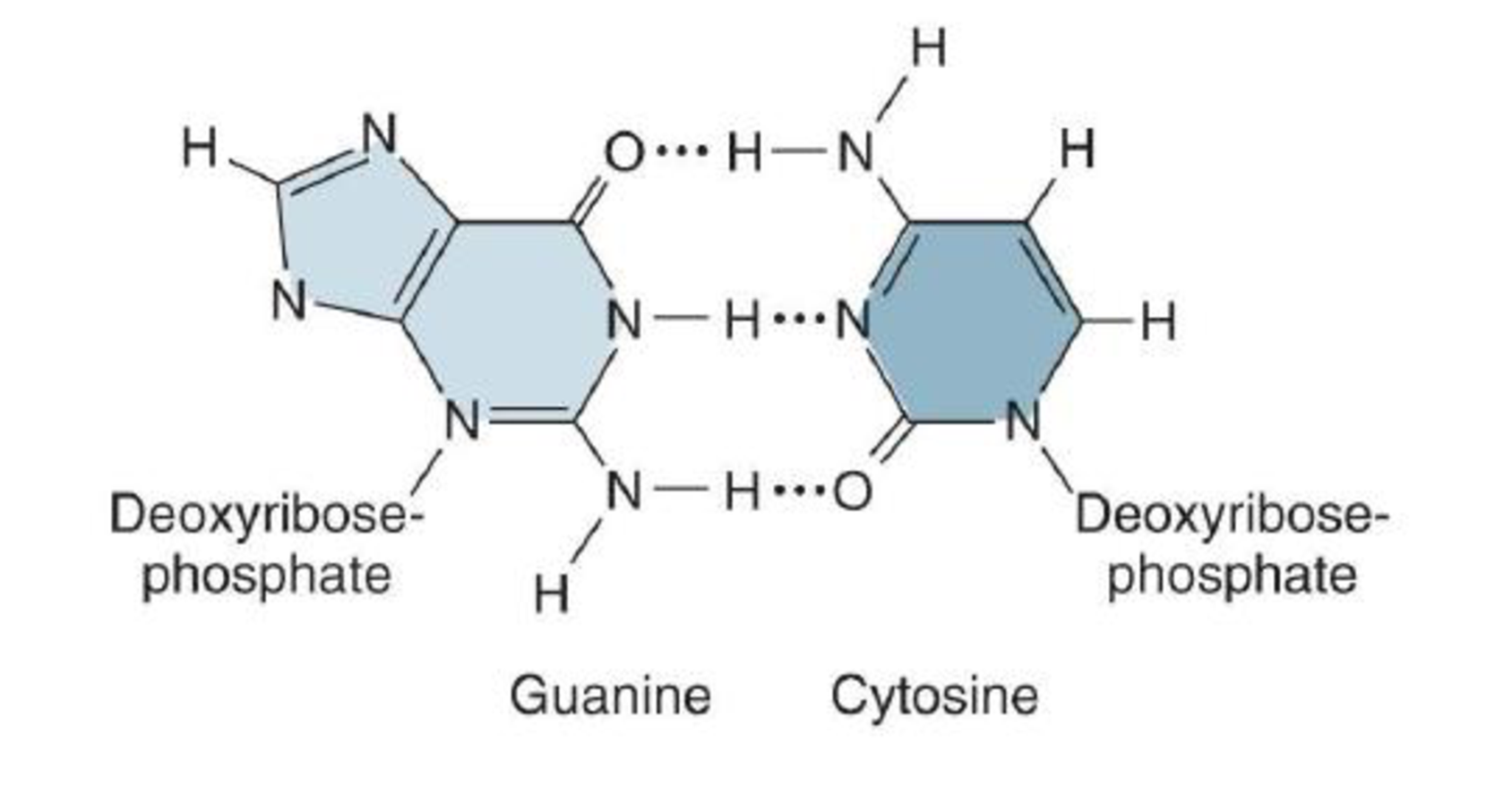
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The carbon-to-nitrogen bond that joins amino acids together is called a.a glycosidic bond. b.a peptide bond. c.a hydrogen bond. d.a double bond.
What type of bonds are formed between complementary bases?
a
triple carbon to carbon bonds
b
hydrogen bonds
c
polar covalent bonds
d
ionic bonds
Which of the following statements is incorrect about disulfide bonds? a. They form between two cysteine amino acids.b. They are noncovalent.c. They contribute to the tertiary structure of some proteins.d. They contribute to the quaternary structure of some proteins.e. They involve the loss of two hydrogen atoms.
Chapter 2 Solutions
Microbiology: An Introduction
Ch. 2 - What is a chemical element?Ch. 2 - DRAW IT Diagram the electronic configuration of a...Ch. 2 - What type of bond holds the following atoms...Ch. 2 - Classify the following types of chemical...Ch. 2 - Bacteria use the enzyme urease to obtain nitrogen...Ch. 2 - Classify the following as subunits of either a...Ch. 2 - DRAW IT The artificial sweetener aspartame, or...Ch. 2 - DRAW IT The following diagram shows the...Ch. 2 - Prob. 9RCh. 2 - Prob. 10R
Ch. 2 - Assume E. coli bacteria are grown in a nutrient...Ch. 2 - If Pseudomonas bacteria are supplied with...Ch. 2 - If E. coli were grown in a medium containing the...Ch. 2 - Prob. 4MCQCh. 2 - Prob. 5MCQCh. 2 - Prob. 6MCQCh. 2 - The dissociation products of the molecules are...Ch. 2 - Prob. 8MCQCh. 2 - The dissociation products of the molecules are...Ch. 2 - Prob. 10MCQCh. 2 - When you blow bubbles into a glass of water, the...Ch. 2 - Prob. 2ACh. 2 - Prob. 3ACh. 2 - Prob. 4ACh. 2 - Prob. 1CAECh. 2 - Prob. 2CAECh. 2 - Newborn babies are tested for phenylketonuria...Ch. 2 - The antibiotic amphotericin B causes leaks in...Ch. 2 - Prob. 5CAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- The monomers that make up proteins are called_____. a. nucleotides b. disaccharides c. amino acids d. chaperonesarrow_forwardWhich type of bond represents a weak chemical bond? a. hydrogen bond b. ionic bond c. covalent bond d. polar covalent bondarrow_forward_______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) c. Methyl (CH3) b. Carboxyl (COOH) d. Phosphate (PO4)arrow_forward
- Fatty acids that have ______ are more likely to be liquid at room temperature. A. more double bonds B. fewer double bonds C. a longer carbon chain D. a shorter carbon chainarrow_forwardWhich of the following is incorrect regarding nucleotides?a. They contain a sugar, a nitrogen-containing base, and aphosphate group.b. They are the monomers of fats and polysaccharides.c. They join together by alternating covalent bonds between thesugars and phosphate groups.d. They are present in both DNA and RNAarrow_forwardCompare nucleic acids with polysaccharides. Which of the following is NOT true? Group of answer choices A) Both have monomers that contain simple sugars. B) Both function as structural support for cells. C) Both types of molecules are organic. D) Both are polymerized by dehydration reactions. .arrow_forward
- Polyunsaturated fatty acids a). are solid at room temperature b). have carbon-carbon double bonds c). are polar d). have high melting pointsarrow_forwardDefine the following terms: a. carbonyl group b. carboxyl group c. amino group d. hydroxyl group e. peptide bondarrow_forwardWhich of the following is the characteristic that is common to all lipids?* A. They are made up of lots of double bonds B. They are acidic when mixed with water C. They are insoluble in water D. They do not have high energy contentarrow_forward
- _________________ are the building blocks of proteins. a. Nucleic Acids b. Amino Acids c. Hydrochloric Acids d. Ribonucleic Acidsarrow_forwardWhich type of bond is formed through interactions of partial charges? A. Ionic B. Covalent C. Hydrogen D. Peptidearrow_forwardHow many amino acids and peptide bonds does a tetrapeptide have? A) four amino acids and three peptide bonds B) four amino acids and four peptide bonds C) three amino acids and four peptide bonds D) four amino acids and two peptide bondsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College

Biology (MindTap Course List)
Biology
ISBN:9781337392938
Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:Cengage Learning

GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license