
BIOLOGY:CONCEPTS...LL
null Edition
ISBN: 9781260867770
Author: Hoefnagels
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 4PIT
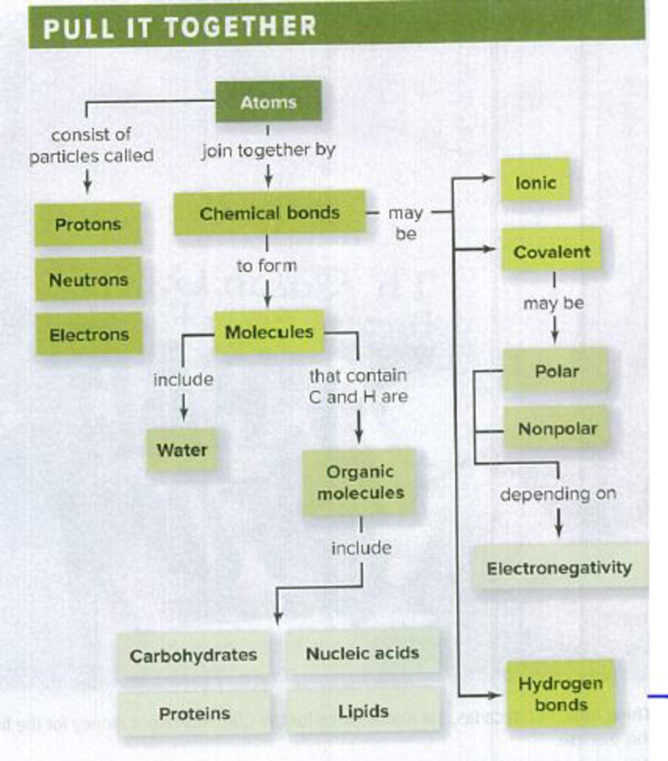
4. Add monomers, polymers, dehydration synthesis, and hydrolysis to this concept map.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Explain using words and diagrams the structure of carbohydrates
2. Demonstrate understanding of monomers and polymers in car
1. Identify and encircle the functional groups in the structure of molecule
1. Explain why lipids are insoluble in polar solvents.
2. How do oils and fats differ?
Chapter 2 Solutions
BIOLOGY:CONCEPTS...LL
Ch. 2.1 - Which four elements do organisms require in the...Ch. 2.1 - Where in an atom are protons, neutrons, and...Ch. 2.1 - What does an elements atomic number indicate?Ch. 2.1 - What is the relationship between the mass of 12C...Ch. 2.1 - Prob. 5MCCh. 2.2 - How are atoms, molecules, and compounds related?Ch. 2.2 - Prob. 2MCCh. 2.2 - Explain how electronegativity differences between...Ch. 2.3 - Prob. 1MCCh. 2.3 - Distinguish between a solute and a solvent and...
Ch. 2.3 - Prob. 3MCCh. 2.3 - Prob. 4MCCh. 2.3 - Prob. 5MCCh. 2.3 - Prob. 6MCCh. 2.4 - How do acids and bases affect a solution's H'...Ch. 2.4 - Prob. 2MCCh. 2.4 - How do buffers regulate the pH of a fluid?Ch. 2.5 - Prob. 1MCCh. 2.5 - Prob. 2MCCh. 2.5 - List examples of carbohydrates, proteins, nucleic...Ch. 2.5 - What is the significance of a protein's shape, and...Ch. 2.5 - What are some differences between RNA and DNA?Ch. 2.5 - Prob. 6MCCh. 2.6 - Prob. 1MCCh. 2.6 - Prob. 2MCCh. 2 - A hydrogen ion (H+) has...Ch. 2 - Prob. 2MCQCh. 2 - Prob. 3MCQCh. 2 - Prob. 4MCQCh. 2 - Prob. 5MCQCh. 2 - Prob. 6MCQCh. 2 - The vitamin biotin contains 10 atoms of carbon, 16...Ch. 2 - Distinguish between nonpolar covalent bonds, polar...Ch. 2 - Prob. 3WIOCh. 2 - Prob. 4WIOCh. 2 - Define solute, solvent, and solution.Ch. 2 - Give an example from everyday life of each of the...Ch. 2 - Prob. 7WIOCh. 2 - How do hydrogen ions relate to the pH scale?Ch. 2 - Sketch a monosaccharide, an amino acid, a...Ch. 2 - Prob. 10WIOCh. 2 - You eat a sandwich made of starchy bread, ham, and...Ch. 2 - Prob. 1PITCh. 2 - Prob. 2PITCh. 2 - Besides water, what other molecules are essential...Ch. 2 - 4. Add monomers, polymers, dehydration synthesis,...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Propose a model for the assembly of a flagellum in a typical Gram-positive cell envelope.
Prescott's Microbiology
Relative thickness of the myocardium in different chambers; the functional significance of those differences; a...
Anatomy & Physiology: The Unity of Form and Function
Some people consider Pasteur or Koch to be the Father of Microbiology, rather than Leeuwenhoek. Why might they ...
Microbiology with Diseases by Body System (5th Edition)
Visit this site (http://openstaxcollege.org/l/heartvalve) to observe an echocardiogram of actual heart valves o...
Anatomy & Physiology
6. How can you use the features found in each chapter?
Human Anatomy & Physiology
11. In the early 1800s, French naturalist Jean Baptiste Lamarck suggested that the best explanation for the rel...
Campbell Biology: Concepts & Connections (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- During the breakdown of polymers, which of the following reactions takes place? hydrolysis dehydration condensation covalent bondarrow_forwardWhich of the following characteristics is not common to carbohydrates, lipids, and proteins? a. They are composed of a carbon backbone with functionalgroups attached. b. Monomers of these molecules undergo dehydration synthesisto form polymers. c. Their polymers are broken apart by hydrolysis. d. The backbones of the polymers are primarily polar molecules. e. The molecules are held together by covalent bonding.arrow_forwardThe characteristic that gives an element its distinctive properties is its number of ________. protons neutrons electrons atomsarrow_forward
- The mutual attraction of opposite charges holds atoms together as molecules in a(n) ________ bond. a. ionic b. hydrogen c. polar covalent d. nonpolar covalentarrow_forwardOrganic molecules consist mainly of ______ atoms. a. carbon b. carbon and oxygen c. carbon and hydrogen d. carbon and nitrogenarrow_forwardThe water lattice: a. is formed from hydrophobic bonds. b. causes ice to be denser than water. c. causes water to have a relatively low specific heat. d. excludes nonpolar substances. e. is held together by hydrogen bonds that are permanent; that is, they never break and reform.arrow_forward
- Which of the following is not a property of water? a. It has a low boiling point compared with other molecules. b. It has a high heat of vaporization. c. Its molecules resist separation, a property called cohesion. d. It has the property of adhesion, the ability to stick to chargedand polar groups in molecules. e. It can form hydrogen bonds to molecules below but not aboveits surface.arrow_forwardWhich functional group has a double bond and forms organic acids? a. carboxyl b. amino c. hydroxyl d. carbonyl e. sulfhydrylarrow_forwardWhen electrons are shared equally between atoms, they form: a. a polar covalent bond. b. a nonpolar covalent bond. c. an ionic bond. d. a hydrogen bond. e. a van der Waals force.arrow_forward
- The measure of an atoms ability to pull electrons away from another atom is called ________ . a. electronegativity b. charge c. polarityarrow_forwardThe measure of an atoms ability to pull electrons away from another atom is called ________ . a. electronegativity b. charge c. polarityarrow_forwardWhat atom has only one proton? a. hydrogen b. an isotope c. a free radical d. a radioisotopearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Biology: The Unity and Diversity of Life (MindTap...
Biology
ISBN:9781337408332
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Anatomy & Physiology
Biology
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:OpenStax College

Biology: The Unity and Diversity of Life (MindTap...
Biology
ISBN:9781305073951
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Cengage Learning
What is Metabolism?; Author: Stated Clearly;https://www.youtube.com/watch?v=nRq6N5NGD1U;License: Standard youtube license