
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 20, Problem 1Q
Which of the following would be soluble in dilute aqueous HCl?
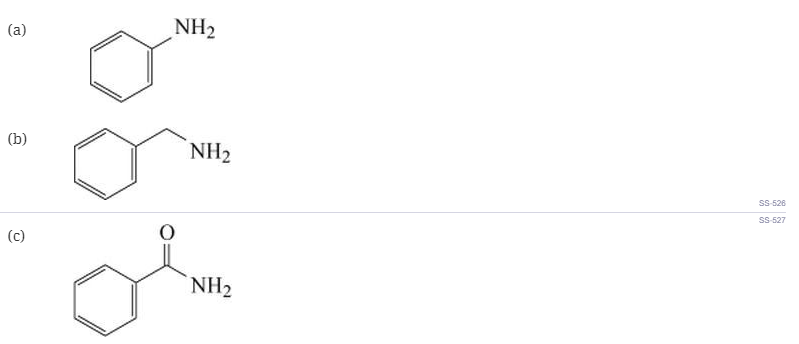
(d) Two of the above
(e) All of the above
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
which one of the following is least soluble in water?
which of the following gives the correct order of solubility?
Please do it correctly and neat and clean
Fill out Table 1 above by writing “+” if the compound is soluble/miscible in the solvent. Otherwise, write “-“.
Chapter 20 Solutions
Organic Chemistry
Ch. 20 - Prob. 1PPCh. 20 - Prob. 2PPCh. 20 - Prob. 3PPCh. 20 - PRACTICE PROBLEM 20.5 Outline a preparation of...Ch. 20 - Prob. 5PPCh. 20 - Prob. 6PPCh. 20 - Prob. 7PPCh. 20 - Prob. 8PPCh. 20 - Prob. 9PPCh. 20 - Practice Problem 20.11 In the preceding examples...
Ch. 20 - Prob. 11PPCh. 20 - Prob. 12PPCh. 20 - Practice Problem 20.14
Outline a synthesis of...Ch. 20 - Prob. 14PPCh. 20 - Prob. 15PPCh. 20 - PRACTICE PROBLEM
20.16
An amine A has the...Ch. 20 - PRACTICE PROBLEM Sulfonamides of primary amines...Ch. 20 - PRACTICE PROBLEM
20.18 (a) Starting with aniline...Ch. 20 - Prob. 19PCh. 20 - 20.20 Give common or systematic names for each of...Ch. 20 - Which is the most basic nitrogen in each compound?...Ch. 20 - Prob. 22PCh. 20 - Prob. 23PCh. 20 - Show how you might synthesize each of the...Ch. 20 - Prob. 25PCh. 20 - 20.26 Provide the major organic product from each...Ch. 20 - Prob. 27PCh. 20 - Prob. 28PCh. 20 - Prob. 29PCh. 20 - Prob. 30PCh. 20 - Prob. 31PCh. 20 - Write equations for simple chemical rests or state...Ch. 20 - Prob. 33PCh. 20 - 20.34 Using reactions that we have studied in this...Ch. 20 - 20.35 Provide a detailed mechanism for each of the...Ch. 20 - Prob. 36PCh. 20 - Prob. 37PCh. 20 - Prob. 38PCh. 20 - Prob. 39PCh. 20 - 20.40 Give structures for compounds R-W:
Ch. 20 - Prob. 41PCh. 20 - Prob. 42PCh. 20 - Diethylpropion (shown here) is a compound used in...Ch. 20 - Prob. 44PCh. 20 - 20.45 Compound W is soluble in dilute aqueous HCI...Ch. 20 - 20.46 Propose structures for compounds X, Y, and...Ch. 20 - Compound A(C10H15N) is soluble in dilute HCI. The...Ch. 20 - Prob. 48PCh. 20 - Prob. 49PCh. 20 - 20.52 When phenyl isochiocyanatc, , is reduced...Ch. 20 - Prob. 51PCh. 20 - 20.54 Propose a mechanism that can explain the...Ch. 20 - When acetone is treated with anhydrous ammonia in...Ch. 20 - Prob. 54PCh. 20 - Which of the following would be soluble in dilute...Ch. 20 - Which would yield propylamine? (d) Two of the...Ch. 20 - Select the reagent from the list below that could...Ch. 20 - Prob. 4QCh. 20 - 20.5 Select the stronger base from each pair (in...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Reason for table salt, NaCl, to not have a smell needs to be determined. Concept Introduction : Molecules smell...
Living by Chemistry
Look up the physical properties of several of the compounds you will be making in your lab from several differe...
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
17.33 On heating, cis-4-hydroxycyclohexanecarboxylic acid forms a lactone but trans-4-hydroxycyclohexanecarboxy...
Organic Chemistry
In qualitative analysis, Ca2+ and Ba2+ are separated from Na+, K+, and Mg2+ by adding aqueous (NH4)2CO3 to a so...
General Chemistry: Atoms First
a. How can aspirin be synthesized from benzene? b. Ibuprofen is the active ingredient in pain relievers such as...
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Figure 16.3 shows the solubility of AgNO3 in water inunits of moles of AgNO3 per kilogram of H2O. If 255 g of AgNO3 is added to 100 g of water at 95°C and cooledslowly, at what temperature will the solution becomesaturated?arrow_forwardpredict which is more solublearrow_forwardExplain why isopropanol is soluble in conc Sulfuric acidarrow_forward
- A 0.9% glucose solution is prepared by diluting __ul of a 0.06M solution in 1.8ml of water with lots of explanation pleasearrow_forwardWould you expect the given compund to be more soluble in 0.1M strong acid or 0.1M strong base solution. Justify by indicating the appropriate functional groups responsible for increased solubility.arrow_forwardConsider the following salts. Which is the MOST soluble in water? Explain your answer.arrow_forward
- Thymol is very slightly soluble in water and very soluble in 1.0 M NaOH. Explain.arrow_forwardWhich of the following is expected to be less soluble in water: ZnS or HgS? Explain the answer in detailarrow_forwardGive typed full explanation Consideran aqueous solution at a particular temperature where the rate of crystallization is that same as the rate of dissolution. What is true? Assume that there is solid at the bottom of the beaker The solution is unsaturated, and no more solute can dissolve. The solution is saturated, and no more solute can dissolve. The solution is unsaturated, and more solute can dissolve The solution is saturated, and more solute can dissolve.arrow_forward
- Q1. How will you compare the boiling temperature of the solutions with that of a pure water? Q2. Does sodium chloride seem to deviate more from the boiling temperature and freezing temperature of pure water than the sucrose and glycerol solutions? Why?arrow_forwardWhy is maleic acid (a polar compound) partially soluble in hexane (a nonpolar compound)?arrow_forwardDescribe in detail the steps involved to prepare the solution below. a) 0.1 Molal Mohar' Salt. Note please show all the steps. Make it lengthy.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Intermolecular Forces and Boiling Points; Author: Professor Dave Explains;https://www.youtube.com/watch?v=08kGgrqaZXA;License: Standard YouTube License, CC-BY