
Concept explainers
(a)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond
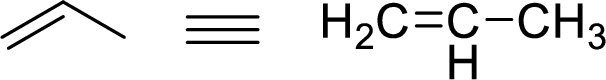
Stability of the allylic carbocations:
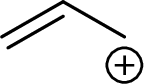
Example for Primary allylic carbocation is:
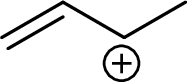
Example for Secondary allylic carbocation is:
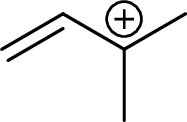
Example for tertiary allylic carbocation is:
(b)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond alkene functional group as shown here:
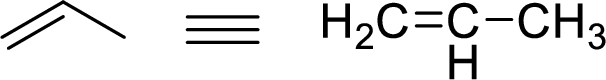
Stability of the allylic carbocations:
Example for Primary allylic carbocation is:
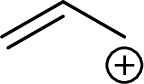
Example for Secondary allylic carbocation is:
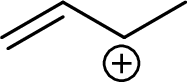
Example for tertiary allylic carbocation is:
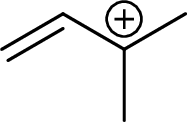
(c)
Interpretation: The important resonance hybrids for the given allylic carbocations have to be drawn and ranked based on their contributions.
Concept Introduction:
Allylic systems:
The allylic system is characterized by the connectivity of a single bonded carbon atom connected to double bond alkene functional group as shown here:
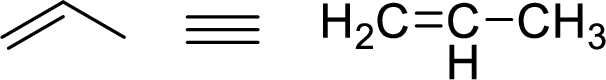
Stability of the allylic carbocations:
Example for Primary allylic carbocation is:
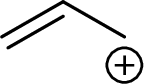
Example for Secondary allylic carbocation is:
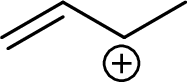
Example for tertiary allylic carbocation is:
Trending nowThis is a popular solution!

Chapter 20 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Write and the major product for each of the following reactions. Are these reactions stereospecific? Why, or why not? Are the reactions Stereoselective? Why or why not?arrow_forwardRank these carbocations from the least stable cation to most.arrow_forwardRank the carbocation compounds below from most stable to least stable.arrow_forward
- addition of hbr to a double bond with an ether (-or) substituent occurs regiospecifically to give a product in which the Br OR are bonded to the same carbon. Draw the two possible carbocation intermediates in this electrophilic addition reaction,and explain using resonance why the observed product is formed.arrow_forwardArrange the following compounds in order of their reactivity from most reactive to least reactive.arrow_forwardWrite an additional resonance contributing structure for each carbocation and state which of the two makes the greater contribution to the resonance hybrid. Classify each additional contributing structure as a 1°, 2°, or 3° allylic cationarrow_forward
- 4) Draw the complete electron-pushing arrow mechanism for the following reductions. Explain, using resonance contributors, the regiochemistry that results in each case. ỌMe Na, MeOH ? NH3 CHO Na, MeOH ? NH3arrow_forwardDraw all resonance structures for the carbocation formed by ortho attack of +NO2 on each starting material. Label any resonance structures that are especially stable or unstable with reasonsarrow_forwardRank the following carbocation in order of increasing stability?arrow_forward
- Resveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inflammatory, and various cardiovascular effects are under active investigation. (a) Draw all resonance structures for the radical that results from homolysis of the OH bond shown in red. (b) Explain why homolysis of this OH bond is preferred to homolysis of either OH bond in the other benzene ring.arrow_forwardDehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3 CHCCH3 OH CH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodlearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
