
Interpretation: In the given intramolecular Diels-Alder reaction, the structural formula of the product has to be predicted.
Concept Introduction:
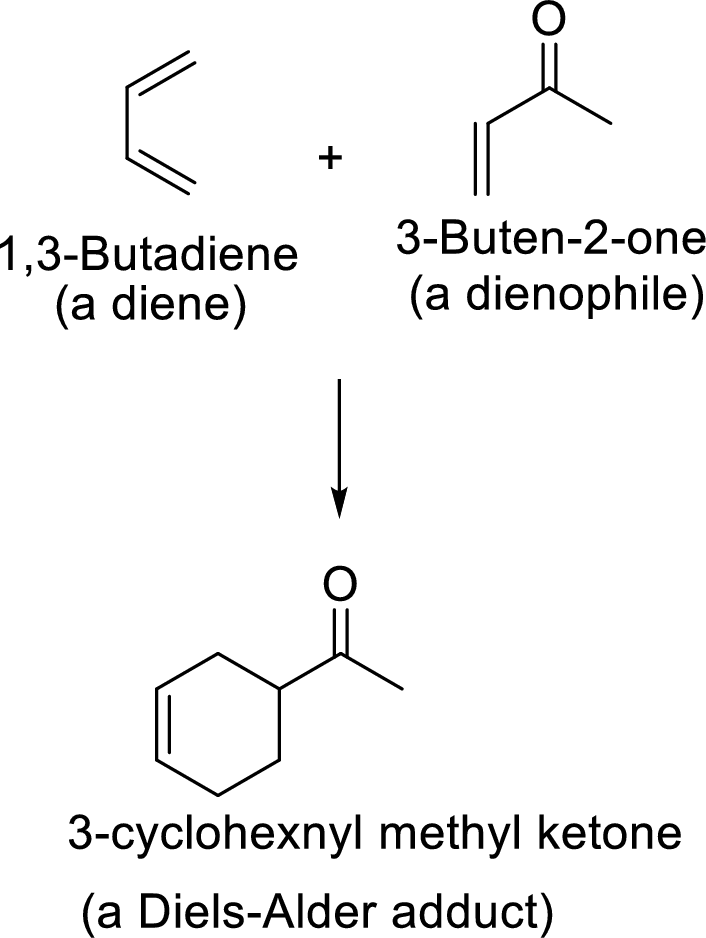
Diels-Alder reaction:
It is the reaction of conjugated dienes with double or triple bonded compounds which are known as “dienophiles”. The reaction is a
Example:
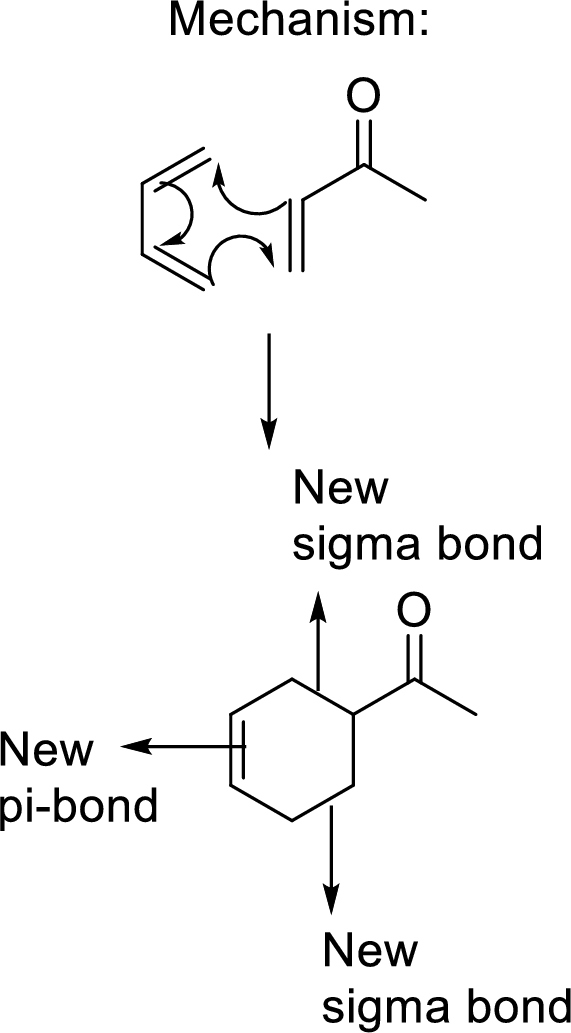
This mechanism shown that three
Diels-Alder reaction to form bicyclic system:
The Diels-Alder adduct formed in the Diels-Alder reaction can also be a bicyclic system which will be obtained when cylopentadiene is used as the diene as shown here:
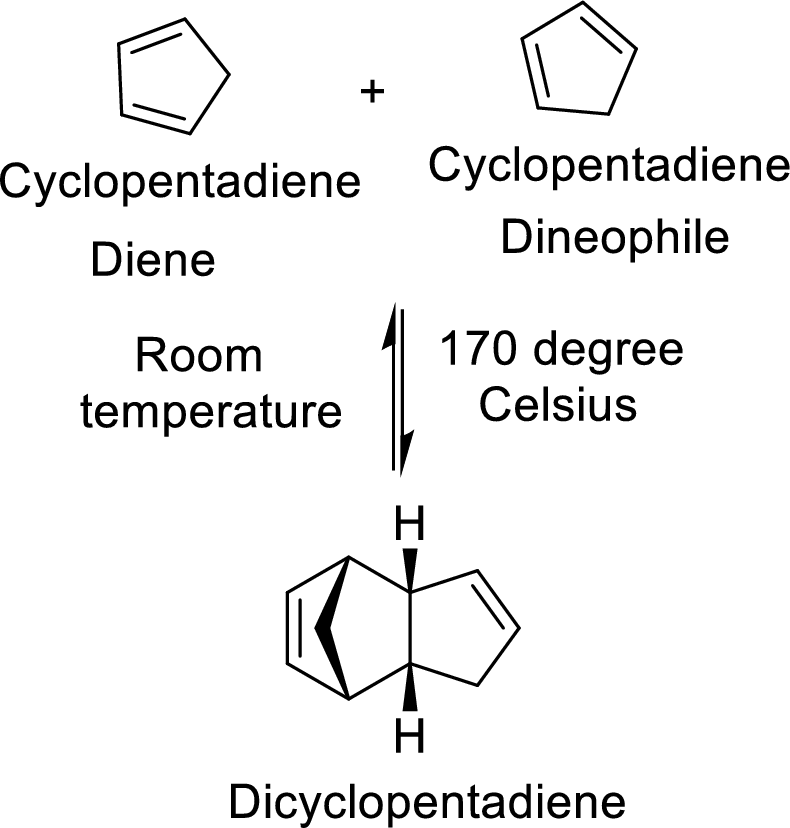
In this reaction, the cylopentadiene acts as both diene and dienophile and formed a bicyclic system. When it is heated to
Intramolecular reaction:
In the word “Intramolecular”- “Intra”- means “within” or “in”. So an intramolecular reaction means the reaction is taking place within one molecule itself without the involvement of any other molecules.
Trending nowThis is a popular solution!

Chapter 20 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardWhen 1,2-epoxycyclohexane (cyclohexene oxide) is treated with anhydrous HCl inmethanol, the principal product is trans-2-methoxycyclohexanol. Propose a mechanismto account for the formation of this product.arrow_forwardShow how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forward
- When the following compound undergoes solvolysis in ethanol, three products are obtained. Propose a mechanism to account for the formation of these products.arrow_forwardTreatment of a hydrocarbon A (molecular formula C9H18) with Br2 in the presence of light forms alkyl halides B and C, both having molecular formula C9H17Br. Reaction of either B or C with KOC(CH3)3 forms compound D (C9H16) as the major product. Ozonolysis of D forms cyclohexanone and acetone. Identify the structures of A–D.arrow_forwardWhen cyclohexene is treated with bromine in saturated aqueous sodium chloride, a mixture of trans-2-bromocyclohexanol and trans-1-bromo-2-chlorocyclohexane results.Propose a mechanism to account for these two products.arrow_forward
- The following reaction was performed as part of a research program sponsored by the National Institutes of Health to develop therapeutic agents for the treatment of cocaine addiction. Using what you have seen about the reactions of halogens with alkenes, propose a mechanism for this process.arrow_forwardClaisen rearrangement of an allyl phenyl ether with substituent groups in both ortho positions leads to the formation of a para-substituted product. Propose a mechanism for the following rearrangement.arrow_forwardBicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forward
- When heated, allyl aryl ethers and allyl vinyl ethers undergo a reaction called a Claisen rearrangement, a concerted reorganization of bonding electrons similar to the Diels-Alder reaction. The reaction proceeds through a six-membered, cyclic transition state. Draw the structure of the expected product when this compound undergoes a Claisen rearrangement.arrow_forwardTreating cyclohexene with HBr in the presence of acetic acid gives bromocyclohexane(85%) and cyclohexyl acetate (15%).Propose a mechanism for the formation of the latter productarrow_forwardThe following product is formed by an intramolecular Diels-Alder reaction. Provide the structure of the starting compound.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

