
Review the oxidation reactions using
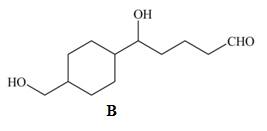
a.
b.
c. PCC
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
ORGANIC CHEM W/CNCT+SOLUTION MANUAL >BI<
- This is a question about balancing oxidation reduction reactions in CHM-152.arrow_forwardPertinent General Reactions. Bayer’s oxidation: Chomic acid oxidation: Lucas test: Ferric Chloride Test:arrow_forward6) What is PCC? Show how you make PCC with CrO3, HCl and Pyridine Why you cannot oxidize 3 ̊-OH with PCC?arrow_forward
- -The solubility of Ag3PO4 is measured and found to be 1.99×10-3 g/L. Use this information to calculate a Ksp value for silver phosphate.Ksp = -The solubility of Zn(CN)2 is measured and found to be 1.45×10-2 g/L. Use this information to calculate a Ksp value for zinc cyanide. Ksp = Please help. Thank you.arrow_forward7) Identify the pair of compounds havingunderlined atoms with the same oxidation number. a) Hg2Cl2 and CaSO4 b) H3PO4 and KMnO4 c) HClO4 and H2Cr2O7 d) LiNO3 and (NH4)3PO4arrow_forwardWhich compound is most likely to undergo oxidative addition of H2? a. RhCl(PPh3)3 b. Fe(CO)5 c. [RhI4(CO)2]− d. [HFe(CO)4]−arrow_forward
- 4. You have a test tube containing a mixture of Ag+, Fe3+, and Ba2+. Make a diagram similar to Expt. 1 to separate the given mixture of cations. Include the correct reagents to be used to effectively isolate cations from each other.arrow_forwardGive equations and name the products for question 8,9,10arrow_forwardComplete the reactions for Group I cations below and answer the questions: (Note: Balance your equations!) Pb2+ + Cl- Ag+ + Cl- Hg22+ + Cl- Pb2+ + I- What is the color of the product from this reaction? Which chloride salt (from Group I cations) dissolves in aqueous NH3? Write the equation for this reaction. Which chloride salt (from Group I cations) is soluble in hot but not cold water?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


