
Organic Chemistry - Access (Custom)
4th Edition
ISBN: 9781259355844
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20, Problem 20.70P
Design a synthesis of each compound from alcohols having four carbons or fewer as the only organic starting materials. Youmay use any other inorganic reagents you choose.
a. 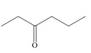 b.
b. ![]() c.
c. 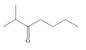 d.
d. 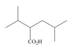 e.
e.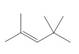
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form asubstituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that canbe used as a synthetic detergent to clean away dirt.
Devise a synthesis of attached compound from the indicated starting material. You may use any other needed organic or inorganic reagents.
Devise a synthesis of each compound from benzene. You may use any other organic or in organic reagents.
Chapter 20 Solutions
Organic Chemistry - Access (Custom)
Ch. 20 - Prob. 20.1PCh. 20 - Which carbonyl groups in the anticancer drug taxol...Ch. 20 - Prob. 20.3PCh. 20 - Prob. 20.4PCh. 20 - Problem 20.5 What aldehyde or ketone is needed to...Ch. 20 - Prob. 20.6PCh. 20 - Problem 20.7 Draw the products formed when is...Ch. 20 - Prob. 20.8PCh. 20 - Prob. 20.9PCh. 20 - Prob. 20.10P
Ch. 20 - Draw the structure of both an acid chloride and an...Ch. 20 - Problem 20.12 Draw the products formed from ...Ch. 20 - Prob. 20.13PCh. 20 - Prob. 20.14PCh. 20 - What product is formed when...Ch. 20 - Prob. 20.16PCh. 20 - Problem-20.16 Review the oxidation reactions using...Ch. 20 - Problem-20.17 Write the step(s) needed to convert ...Ch. 20 - Problem-20.18 Oct-1-yne reacts rapidly with ,...Ch. 20 - Draw the product formed when each organometallic...Ch. 20 - Draw the product of each reaction. a.c. b.d.Ch. 20 - Draw the products including stereochemistry of the...Ch. 20 - What Grignard reagent and carbonyl compound are...Ch. 20 - Problem 20.24 Linalool (the Chapter 9 opening...Ch. 20 - Problem 20.25 What Grignard reagent and carbonyl...Ch. 20 - Prob. 20.26PCh. 20 - Draw the products formed when each compound is...Ch. 20 - What ester and Grignard reagent are needed to...Ch. 20 - What organocuprate reagent is needed to convert...Ch. 20 - What reagent is needed to convert (CH3)2CHCH2CHO...Ch. 20 - Prob. 20.31PCh. 20 - What carboxylic acid formed from each alkyl halide...Ch. 20 - Prob. 20.33PCh. 20 - Prob. 20.34PCh. 20 - Problem 20.35 Synthesize each compound from...Ch. 20 - Prob. 20.36PCh. 20 - 20.37 Devise a synthesis of each alcohol from...Ch. 20 - 20.38 Draw the products formed when pentanal is...Ch. 20 - Prob. 20.39PCh. 20 - Draw the product formed when CH3CH2CH2MgBr is...Ch. 20 - Draw the product formed when (CH3CH2CH2CH2)2CuLi...Ch. 20 - The stereochemistry of the products of reduction...Ch. 20 - Prob. 20.43PCh. 20 - What reagent is needed to convert...Ch. 20 - What reagent is needed to convert...Ch. 20 - Draw the products or each reduction reaction. a....Ch. 20 - Prob. 20.47PCh. 20 - Draw all stereoisomers formed in each reaction. a....Ch. 20 - Prob. 20.49PCh. 20 - 20.46 Treatment of ketone A with ethynylithium...Ch. 20 - 20.47 Explain why metal hydride reduction gives an...Ch. 20 - Prob. 20.52PCh. 20 - 20.49 Identify the lettered compounds in the...Ch. 20 - Prob. 20.54PCh. 20 - Draw a stepwise mechanism for each reaction. a. b.Ch. 20 - Prob. 20.56PCh. 20 - 20.54 Draw a stepwise mechanism for the following...Ch. 20 - What Grignard reagent and aldehyde or ketone are...Ch. 20 - Prob. 20.59PCh. 20 - What ester and Grignard reagent are needed to...Ch. 20 - What organolithium reagent and carbonyl compound...Ch. 20 - 20.59 What epoxide and organometallic reagent are...Ch. 20 - Prob. 20.63PCh. 20 - 20.61 Propose two different methods to synthesize...Ch. 20 - Synthesize each compound from cyclohexanol using...Ch. 20 - Prob. 20.66PCh. 20 - Prob. 20.67PCh. 20 - Devise three different methods to prepare each...Ch. 20 - Convert benzene into each compound. You may also...Ch. 20 - Design a synthesis of each compound from alcohols...Ch. 20 - Synthesize each compound from the given starting...Ch. 20 - 20.69 An unknown compound A (molecular formula )...Ch. 20 - 20.70 Treatment of compound C (molecular formula )...Ch. 20 - 20.71 Treatment of compound E (molecular formula )...Ch. 20 - 20.72 Reaction of butanenitrile () with methyl...Ch. 20 - 20.73 Treatment of isobutene with forms a...Ch. 20 - Prob. 20.77PCh. 20 - Prob. 20.78PCh. 20 - 20.76 Lithium tri-sec-butylborohydride, also known...Ch. 20 - Prob. 20.80PCh. 20 - Prob. 20.81PCh. 20 - Prob. 20.82PCh. 20 - 20.80 Draw a stepwise mechanism for the following...Ch. 20 - Prob. 20.84P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Devise a synthesis of attached compound from the given starting materials.You may also use organic alcohols having four or fewer carbons, andany organic or inorganic reagents.arrow_forwardDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that can be used as a synthetic detergent to clean away dirtarrow_forwardDevise a synthesis of attached compound. You may use HC=CH, ethyleneoxide, and alkyl halides as organic starting materials and any inorganicreagents.arrow_forward
- Devise a synthesis of attached compound from the indicated startingmaterial. You may also use any organic compounds with one or two carbons and any needed inorganic reagents.arrow_forwardDevise a stepwise mechanism for the following reaction.arrow_forwardComplete these reactions. (a) (b)arrow_forward
- Devise a synthesis of each compound from the indicated starting material. You may use any other needed organic or inorganic reagents.arrow_forwardDevise a synthesis of each compound from benzene. You may use alcohols with one or two carbons and any inorganic reagents.arrow_forwardDevise a synthesis of each compound. You may use HC=CH, ethylene oxide, and alkyl halides as organic starting materials and any inorganic reagents.arrow_forward
- Starting from toluene as the only organic reagent you have and use any other inorganic reagents to perparearrow_forwardDevise a synthesis of attached product from the given starting material.More than one step is required.arrow_forwardDraw a stepwise mechanism for the attached reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single steparrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY