
Interpretation:
The following mechanism, which has the curved arrow omitted, is for another reaction that converts an amide to a
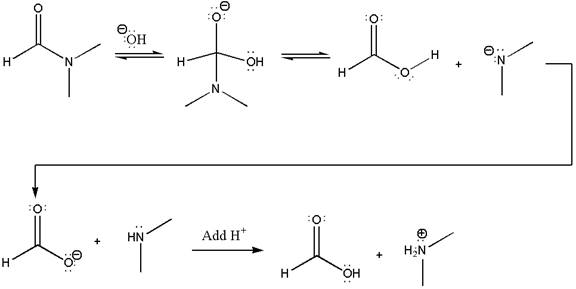
Concept introduction:
The opposite charges attract while the like charges repel. In an elementary step, electrons tend to follow from an electron rich site to an electron poor site. An elementary step can also occur in which only a single bond is broken and both electrons from that bond end up on one of the atoms initially involved in the bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
ORGANIC CHEMISTRY:PRINCIPLES...(PB)
- (Intermediate) Reactant Na+ + Tip: Only add curved arrows in this sketcher Apply Mechanism Hint Solution 1 remaining step(s) can be solvedarrow_forwardDraw the mechanism if it involves general-base catalysis.arrow_forwardDraw the mechanism arrows for the reaction between an acid chloride and an alcohol.arrow_forward
- How many intermediate/s is/are present in the reaction based from the energy diagram as shown belowarrow_forwardShown below is a two-step mechanism beginning with nucleophilic attack of water, and subsequent deprotonation with a base. Draw the arrows for the mechanism for both step 1 and step 2 and draw the intermediate product of in the box. + H-O OH + H₂Oarrow_forwardComplete the reaction map with the choices given.arrow_forward
- Identify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.arrow_forwardDraw the reactant of the following reaction sequence.arrow_forwardWhere do the arrows for this are suppose to go? (Intermediate) Reactant Mechanism Explorer: Sketch and Submission (Intermediate) Reactant Tip: Only add curved arrows in this sketcher Mechanism Explorer: Sketch and Submission (Intermediate) Reactant CH H H₂O Tip: Only add curved arrows in this sketcher learrow_forward
- Complete the following reaction with its mechanism.arrow_forwardFor the following reactions, complete the reactions by giving what is missing. This can be either the starting material, reagent(s) or product. Draw the intermediates for each major steparrow_forwardORGANIC CHEMISTRY: Is this mechanism for dibenzalacetone formation complete? arrows and electrons, and formal charges?. If no, please provide the complete mechanism. From this mechanism in the picture, identify the intermediate benzalacetone.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





