
Concept explainers
(a)
Interpretation:
Hemiacetal C should be identified and that should be label as either a or β anomer.
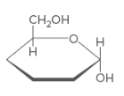
Concept Introduction:
Hemi-acetal is a C atom which connected to two different O atoms. One O atom is in alcohol form and the other one connected to another C atom.
Anomer C-Type of C atoms that are not stereo-genic in linear form, but stereo-genic in cyclic form. stereo-genic center can be classified as either a or β. If the -OH group is located upwards, it is β anomer and if -OH group is located downwards it is a anomer.
(b)
Interpretation:
Hemiacetal C should be identified and that should be label as either a or β anomer.
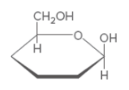
Concept Introduction:
Hemi-acetal is a C atom which connected to two different O atoms. One O atom is in alcohol form and the other one connected to another C atom.
Anomer C-Type of C atoms that are not stereo-genic in linear form, but stereo-genic in cyclic form. stereo-genic center can be classified as either a or β. If the -OH group is located upwards, it is β anomer and if -OH group is located downwards it is a anomer.
(c)
Interpretation:
Hemiacetal C should be identified and that should be label as either a or β anomer.
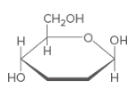
Concept Introduction:
Hemi-acetal is a C atom which connected to two different O atoms. One O atom is in alcohol form and the other one connected to another C atom.
Anomer C-Type of C atoms that are not stereo-genic in linear form, but stereo-genic in cyclic form. stereo-genic center can be classified as either a or β. If the -OH group is located upwards, it is β anomer and if -OH group is located downwards it is a anomer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- Draw the product formed when CH3CH2CH2CH2NH2 reacts with each carbonyl compound in the presence of mild acidarrow_forwardWhat enolate is formed when each ketone is treated with LDA in THF solution? What enolate is formed when these same ketones are treated with NaOCH3 in CH3OH solution?arrow_forwardDraw the product formed when CH3CH2CH2CH2NH2 reacts with each carbonyl compound in the presence of mild acid.arrow_forward
- a.Locate the stereogenic centers in the ball-and-stick model of ezetimibe (trade name Zetia), a cholesterol-lowering drug. b. Label each stereogenic center as R or S.arrow_forwardWhat products are formed when each acetal is hydrolyzed with aqueous acid?arrow_forwardClassify each substituent as electron donating or electron withdrawing.arrow_forward
- Answer the following questions about the carbohydrate erythrulose, an ingredient in sunless tanning agents. erythrulose a. Does erythrulose contain an aldehyde or ketone? b. Classify each hydroxyl group as 1 °, 2 °, or 3 °. c. What product is formed when erythrulose is treated with Tollens reagent? d. What product is formed when erythrulose is treated with K 2Cr 2O 7?arrow_forwardWhat product is formed when each compound is treated with either LiAlH4 (followed by H2O), or NaBH4 in CH3OH?arrow_forwardDraw the products formed (including stereoisomers) when each compound is reduced with NaBH4 in CH3OH.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
