
(a)
Interpretation:
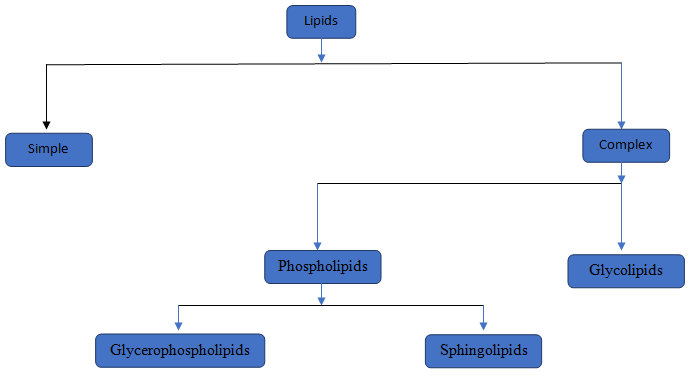
The category to which the given lipid structure belongs should be identified.
Concept Introduction:
Lipids are a type of chemical substances which is not soluble in water but soluble in non-polar solvents. Lipids are of two type simple lipids and complex lipids. Simple lipids are those where Glycerol attached with fatty acid. In complex lipids there are two major lipids i, e, Phospholipids and Glycolipids.
(b)
Interpretation:
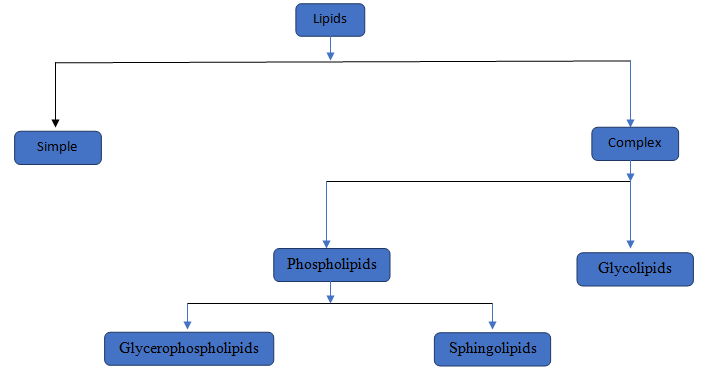
The components of the compound should be determined.
Concept Introduction:
Lipids are a type of chemical substances which is not soluble in water but soluble in non-polar solvents. Lipids are of two type simple lipids and complex lipids. Simple lipids are those where Glycerol attached with fatty acid. In complex lipids there are two major lipids i,e, Phospholipids and Glycolipids.
Trending nowThis is a popular solution!

Chapter 20 Solutions
Introduction To General, Organic, And Biochemistry
- Problem 22-1 Show how to form the dipeptide valyiphenylalanine (Val—Phe).arrow_forwardA protein binds to ligand with ΔG° = -18.3 kJ mol-1 at 37.0 °C. Calculate the Kd at 37 °C. Multiply your answer by 1,000 before entering. (R = 8.3145 J mol-1 K-1)arrow_forwardOCHEM 2 lab What are the amino acids that can give a positive Ceric ammonium nitrate test? Oxidation number of Cerium in the CAN reagent and the final red complex? What are the 2 amino acids (Draw the structures) that can be formed from hydrolysis of aspartame?arrow_forward
- How many moles of Fe(OH)₂ [Ksp = 1.8 x 10-15] will dissolve in 1.0 liter of water buffered at pH=9.92? O2.6x10-7 O 6.9x10-9 O 3.8x106 O 2.2x10-11 O 8.3x10-5 Submit Answer Tryarrow_forwardGlucosidase has KM = 6.0 x 10 -5 M; what value of [S] is needed to get: a) v initial = 0.75 V max b) v initial = 4.0 x 10^-9 M-1 s-1 , given Vmax = 2.0 x 10^-8 M -1 s-1 Please answer fast i give you upvote.arrow_forward1) a) Predict whether fumarate or maleate will have greater electrophoretic mobility. b) Electrophoresis is run with the injection end positive and the detection end negative with a buffer with pH = 8.5. The charge on both ions is -2 at pH = 8.5. The electroosmotic flow is greater than the electrophoretic flow. Predict the order of elution of these two ions. c) At pH = 4.0, both anions have a charge of -1. At this pH, the injection end is negative and the detection end is positive. Why is the polarity switched at lower pH? Predict the order of elution of the two ions.arrow_forward
- The secondary structure of proteins results primarily from which of the following interactions? a. formation of disulfide linkages b. formation of hydrogen bonds c. formation of salt bridges d. more than one response is correct e. no correct response H2 + CH2 = CH2 --> CH3CH3Which of the following could be a substrate for an enzyme that has an induced-fit active site and catalyzes the reaction above?arrow_forwardProblem 24-1 What is the functional difference between G-protein and GTP?arrow_forwardPlz give both answer Thank youarrow_forward
- Can you please explain 7? thank youarrow_forwardFischer Esterification 1) Answer the table. 2) Write the equations involved in the chemical tests. 3) Conclusion Video link: https://drive.google.com/file/d/1wMWprC_667c1B0TH-Dp7HNzQbDqxI6Ba/view?usp=sharingarrow_forwardHello, I'm working on the table for the Absorbance Values of the Cobalt Nitrate Solutions. I'm trying to figure out the Value of k. For Example: Concentration A (1) A (2) Average A Value of k 0.0200M 0.262 0.269 0.3965 ?? 0.400M 0.268 0.311 0.4235 ?? 0.0600M 0.194 0.207 0.2975 0.0800M 0.435 0.462 0.666 Average k value_______________ Thank you,arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
