
Concept explainers
(a)
Interpretation:
The value of
Concept Introduction :
The change in enthalpy
(a)
Answer to Problem 148AE
The value of
Explanation of Solution
The lysine molecules with peptide linkage will be represented as −
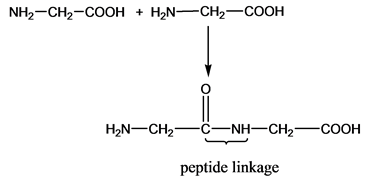
The value of
The values for the bonds which are broken −
Two moles of
Two moles of
Two moles of
Two moles of
Two moles of
Two moles of
Two moles of
Now, the summation of all the above energy will result in the total energy required to break the bonds =
Now, the total energy released when bonds are formed −
Now, the summation of all the above energy will result in the total energy released during the formation of bond =
The value of the energy is positive. Hence, the given reaction is endothermic reaction.
(b)
Interpretation:
The value of
Concept Introduction :
The change in enthalpy
(b)
Answer to Problem 148AE
The value of
Explanation of Solution
The endothermic process can be defined as the process in which heat is absorbed from the surroundings. This process causes the decrement in the entropy of surroundings and it occurs because the molecular motion is also decreased.
Hence, the value of
(c)
Interpretation:
The formation of protein is a spontaneous process or non-spontaneous process is to be predicted.
Concept Introduction :
The change in Gibbs free energy is represented as follows:
Here,
(c)
Answer to Problem 148AE
The synthesis of protein is non-spontaneous process.
Explanation of Solution
The basic equation of the protein formation is given as −
Now, relation between change in Gibbs free energy, change in enthalpy and entropy is given below:
Here, if change in entropy is negative and change in enthalpy is positive thus, change in Gibbs free energy will be positive at all the temperature values.
For positive Gibbs free energy value, the reaction is non-spontaneous.
Want to see more full solutions like this?
Chapter 21 Solutions
Chemical Principles
- In what way is the structure of glycine different from that of the other 19 common amino acids?arrow_forwardWhat element is always present in proteins that is seldom present in carbohydrates and lipids?arrow_forward. How many possible primary structures exist for a small polypeptide containing four individual amino acids?arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





